모든 사진(1)
About This Item
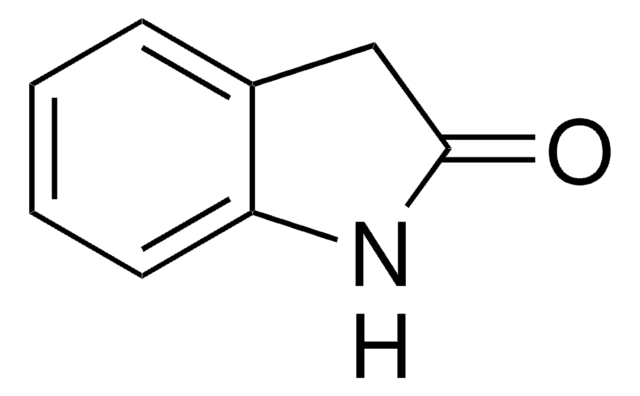
실험식(Hill 표기법):
C8H6ClNO
CAS Number:
Molecular Weight:
167.59
EC Number:
MDL number:
UNSPSC 코드:
12352100
PubChem Substance ID:
NACRES:
NA.22
추천 제품
Quality Level
분석
98%
형태
solid
mp
194-197 °C (lit.)
solubility
methanol: soluble 2.5 mL, clear
작용기
chloro
SMILES string
Clc1ccc2NC(=O)Cc2c1
InChI
1S/C8H6ClNO/c9-6-1-2-7-5(3-6)4-8(11)10-7/h1-3H,4H2,(H,10,11)
InChI key
WWJLCYHYLZZXBE-UHFFFAOYSA-N
일반 설명
5-Chloro-2-oxindole (5-Chlorooxindole) is a starting material for tenidap sodium, a pharmaceutical drug candidate.
애플리케이션
5-Chloro-2-oxindole was used for the quantitation of 5-chloro-2-oxindole, concomitantly with all of its potential positional isomers using a single, highly specific, normal-phase chromatographic system.
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Acute Tox. 4 Oral - Aquatic Chronic 3 - Repr. 2 - Skin Sens. 1
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
S T Colgan et al.
Journal of pharmaceutical and biomedical analysis, 14(7), 825-833 (1996-05-01)
5-Chlorooxindole (5-CO) is a starting material for tenidap sodium, a pharmaceutical drug candidate produced by Pfizer. To insure potency and purity of the drug substance, it is necessary to demonstrate that commercial supplies of 5-CO are free from elevated levels
Boris Letribot et al.
Molecules (Basel, Switzerland), 23(6) (2018-06-13)
Alkylidene oxindoles are important functional moieties and building blocks in pharmaceutical and synthetic chemistry. Our interest in biologically active compounds focused our studies on the synthesis of novel oxindoles, bearing on the exocyclic double bond at the C8, CN, and
Ayman M Saleh et al.
Molecules (Basel, Switzerland), 19(9), 13076-13092 (2014-08-27)
A selected set of substituted pyridone-annelated isoindigos 3a-f has been synthesized via interaction of 5- and 6-substituted oxindoles 2a-f with 6-ethyl-1,2,9-trioxopyrrolo[3,2-f]quinoline-8-carboxylic acid (1) in acetic acid at reflux. Among these isoindigos, the 5'-chloro and 5'-bromo derivatives 3b and 3d show
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.