1.06301
CellPrime® rTrypsin
recombinant Trypsin (powder)
Pharma Manufacturing
Synonym(s):
CellPrime® rTrypsin, Parenzyme
About This Item
Recommended Products
Quality Level
form
powder
pH
5 (20 °C, 10 g/L in H2O)
solubility
10 g/L
bulk density
200 kg/m3
storage temp.
2-8°C
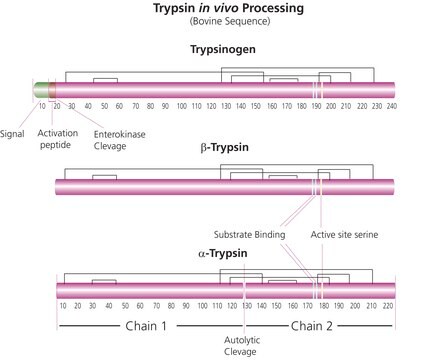
General description
M-Clarity Program
Our comprehensive portfolio of upstream process chemicals not only provides biopharmaceutical manufacturers with high-quality raw materials for production of classical and novel therapies, but also helps them get to market faster and simplify regulatory challenges. Trust us to deliver supply chain transparency and reliable sourcing around the globe, streamlining your product qualification with best-in-class regulatory support and service.
Legal Information
comparable product
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Resp. Sens. 1 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 1
Regulatory Listings
Regulatory Listings are mainly provided for chemical products. Only limited information can be provided here for non-chemical products. No entry means none of the components are listed. It is the user’s obligation to ensure the safe and legal use of the product.
ISHL Indicated Name
Substances Subject to be Indicated Names
ISHL Notified Names
Substances Subject to be Notified Names
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Related Content
Manufacturing viral vaccines is complex and there is no templated approach. Each process must be tailored to the shape, size, nature, physico-chemical behavior, stability, and host specificity of the virus. Learn all about this core platform technology and how expertise—empowered by collaboration— can overcome manufacturing challenges.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service