176176
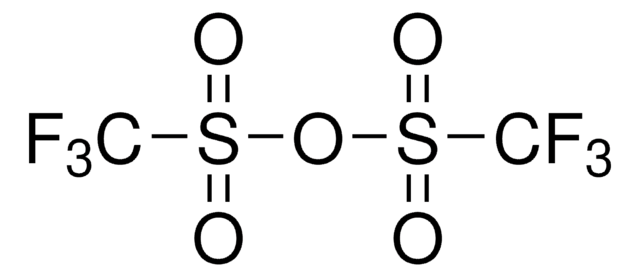
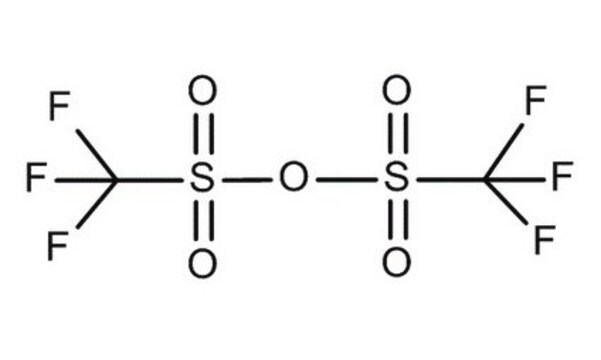
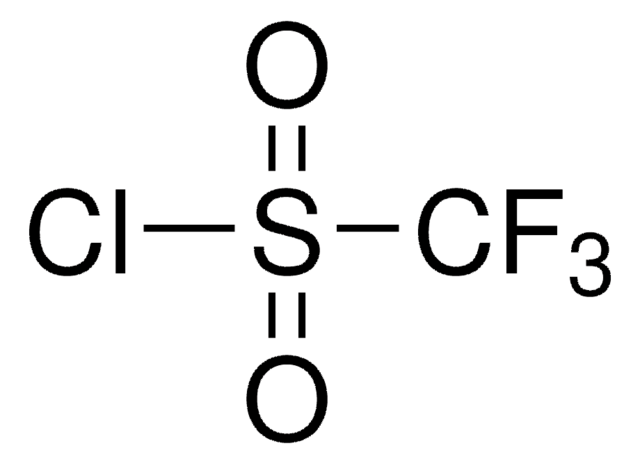
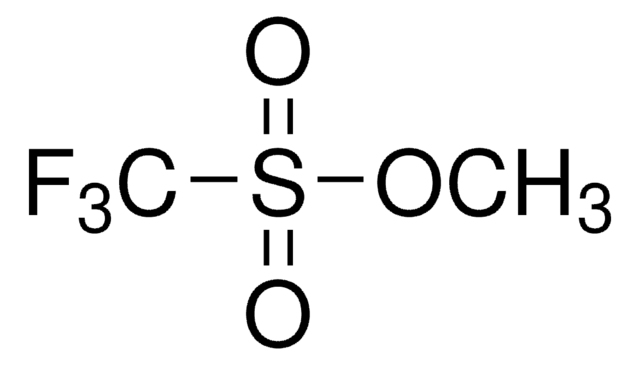
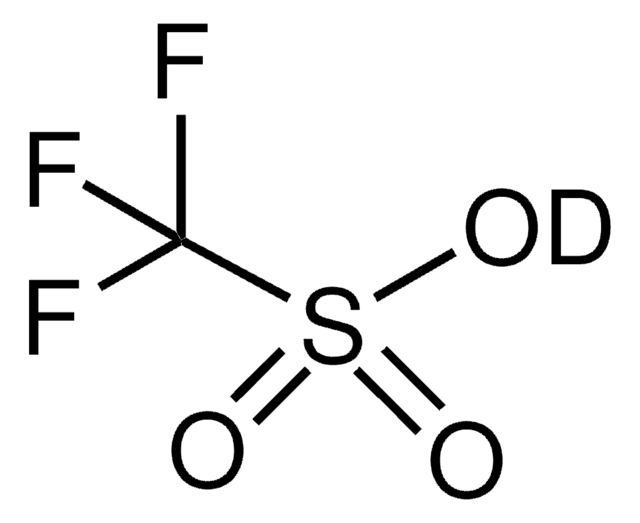
Trifluoromethanesulfonic anhydride
99%
Synonym(s):
Triflic anhydride
About This Item
Recommended Products
vapor density
5.2 (vs air)
Quality Level
vapor pressure
8 mmHg ( 20 °C)
Assay
99%
form
liquid
refractive index
n20/D 1.321 (lit.)
bp
81-83 °C (lit.)
density
1.677 g/mL at 25 °C (lit.)
functional group
fluoro
triflate
SMILES string
FC(F)(F)S(=O)(=O)OS(=O)(=O)C(F)(F)F
InChI
1S/C2F6O5S2/c3-1(4,5)14(9,10)13-15(11,12)2(6,7)8
InChI key
WJKHJLXJJJATHN-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
Reagent for stereoselective synthesis of mannosazide methyl uronate donors
Activator for direct glycosylation with anomeric hydroxy sugars
- A Reactant in the synthesis of Dipentaerythritol hexatriflate via triflatation method and Azido-diphenyl-acetic acid.
- Catalyst for glycosylation for synthesis of polysaccharides.
- Reagent for stereoselective synthesis of mannosazide methyl uronate donors.
- Activator for direct glycosylation with anomeric hydroxy sugars
- A methylation reagent to synthesize trifluoromethylated compounds by direct introduction of CF3 group to (hetero)arenes.
- A reagent to prepare substituted tetrazoles from secondary amides using sodium azide.
- A reagent in Bischler−Napieraiski cyclization reaction along with 4-(N,N-dimethylamnino)pyridine.
accessory
recommended
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Eye Dam. 1 - Ox. Liq. 2 - Skin Corr. 1B - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
5.1B - Oxidizing hazardous materials
WGK
WGK 3
Flash Point(F)
not determinedboils before flash
Flash Point(C)
not determinedboils before flash
Personal Protective Equipment
Regulatory Listings
Regulatory Listings are mainly provided for chemical products. Only limited information can be provided here for non-chemical products. No entry means none of the components are listed. It is the user’s obligation to ensure the safe and legal use of the product.
JAN Code
176176-250G:
176176-500G:
176176-5G:
176176-BULK:
176176-50G:
176176-VAR:
176176-PZ:
176176-2.5KG:
176176-4KG:
176176-20KG:
176176-10G:
176176-1KG:
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service