158534
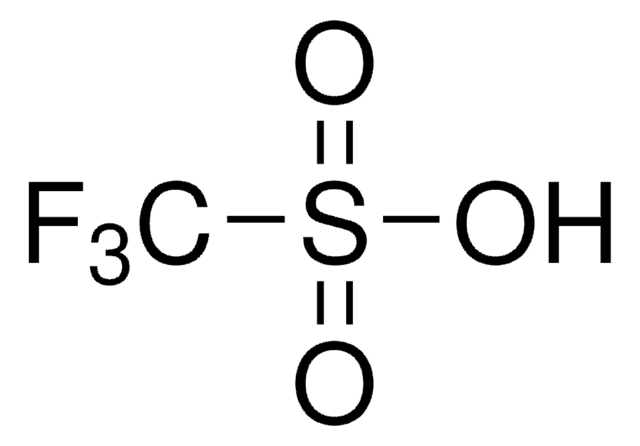
Trifluoromethanesulfonic acid
reagent grade, 98%
Synonym(s):
TFMSA, Triflic acid
About This Item
Recommended Products
grade
reagent grade
Quality Level
vapor density
5.2 (vs air)
vapor pressure
8 mmHg ( 25 °C)
Assay
98%
form
liquid
refractive index
n20/D 1.327 (lit.)
bp
162 °C (lit.)
density
1.696 g/mL at 25 °C (lit.)
functional group
fluoro
triflate
SMILES string
OS(=O)(=O)C(F)(F)F
InChI
1S/CHF3O3S/c2-1(3,4)8(5,6)7/h(H,5,6,7)
InChI key
ITMCEJHCFYSIIV-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
- Friedel-Crafts acylation of aromatic compounds with methyl benzoate.
- Addition reaction of dialkyl disulfides to terminal alkynes.
- Synthesis of a single cyclic tetrasiloxane containing propylammonium trifluoromethanesulfonate and methyl side-chain groups (Am-CyTS).
- Preparation of starting reagents for the synthesis of fluorinated 2,5-substituted 1-ethyl-1H-benzimidazole derivatives.
- Synthesis of aryl triflates, the lactonization of alkenoic acids, and the formation of E-alkenes.
accessory
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Met. Corr. 1 - Skin Corr. 1B - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 1
Flash Point(F)
>332.1 °F - Pensky-Martens closed cup
Flash Point(C)
> 166.7 °C - Pensky-Martens closed cup
Personal Protective Equipment
Regulatory Listings
Regulatory Listings are mainly provided for chemical products. Only limited information can be provided here for non-chemical products. No entry means none of the components are listed. It is the user’s obligation to ensure the safe and legal use of the product.
PDSCL
Deleterious substance
ISHL Indicated Name
Substances Subject to be Indicated Names
ISHL Notified Names
Substances Subject to be Notified Names
JAN Code
158534-VAR:
158534-100G:4548173309514
158534-10G:4548173108315
158534-BULK:
158534-50G:4548173108322
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
The Friedel–Crafts acylation is the reaction of an arene with acyl chlorides or anhydrides using a strong Lewis acid catalyst. This reaction proceeds via electrophilic aromatic substitution to form monoacylated products.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service