21906
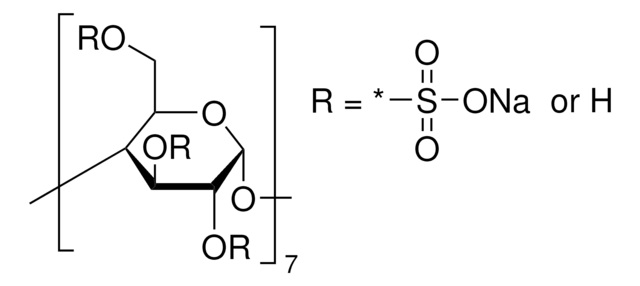
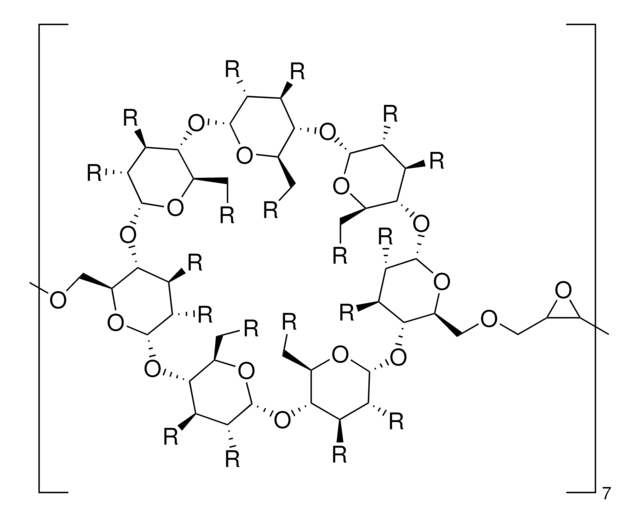
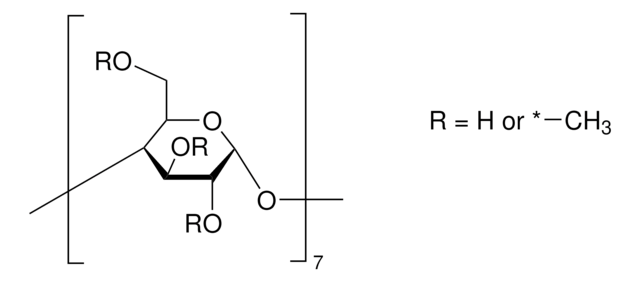
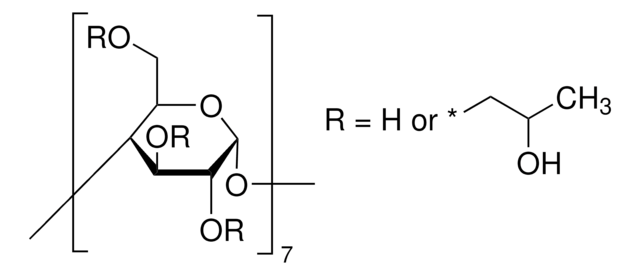
Carboxymethyl-β-cyclodextrin sodium salt
Sinonimo/i:
Sodium CM-β-Cyclodextrin
About This Item
Prodotti consigliati
Forma fisica
solid
Livello qualitativo
Impurezze
10% water
Punto di fusione
~245 °C (dec.)
Solubilità
H2O: 50 mg/mL, clear to slightly hazy, colorless
Stringa SMILE
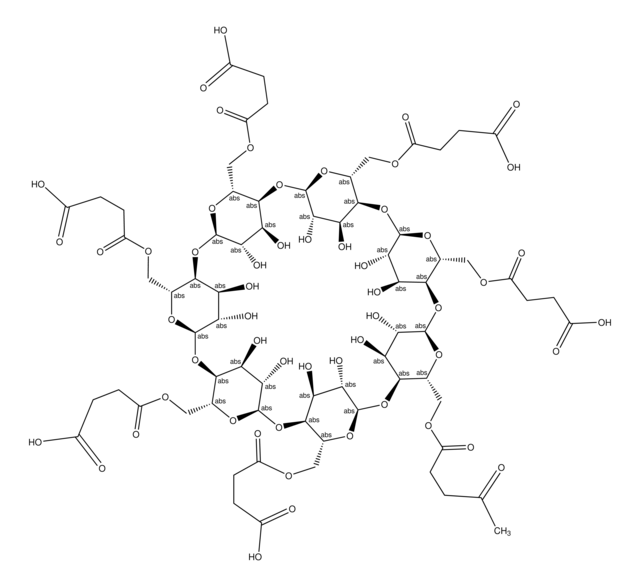
O[C@H]1[C@H](O)[C@@H]2O[C@H]3O[C@H](COCC(O)=O)[C@@H](O[C@H]4O[C@H](COCC(O)=O)[C@@H](O[C@H]5O[C@H](COCC(O)=O)[C@@H](O[C@H]6O[C@H](COCC(O)=O)[C@@H](O[C@H]7O[C@H](COCC(O)=O)[C@@H](O[C@H]8O[C@H](COCC(O)=O)[C@@H](O[C@H]1O[C@@H]2COCC(O)=O)[C@@H](O)[C@@H]8O)[C@@H](O)[C@@H]7O)[C@@H](O)[C@@H]6O)[C@@H](O)[C@@H]5O)[C@@H](O)[C@@H]4O)[C@@H](O)[C@@H]3O
InChI
1S/C56H84O49/c57-22(58)8-85-1-15-43-29(71)36(78)50(92-15)100-44-16(2-86-9-23(59)60)94-52(38(80)31(44)73)102-46-18(4-88-11-25(63)64)96-54(40(82)33(46)75)104-48-20(6-90-13-27(67)68)98-56(42(84)35(48)77)105-49-21(7-91-14-28(69)70)97-55(41(83)34(49)76)103-47-19(5-89-12-26(65)66)95-53(39(81)32(47)74)101-45-17(3-87-10-24(61)62)93-51(99-43)37(79)30(45)72/h15-21,29-56,71-84H,1-14H2,(H,57,58)(H,59,60)(H,61,62)(H,63,64)(H,65,66)(H,67,68)(H,69,70)/t15-,16-,17-,18-,19-,20-,21-,29+,30+,31+,32+,33+,34+,35+,36+,37+,38+,39+,40+,41+,42+,43-,44-,45-,46-,47-,48-,49-,50-,51-,52-,53-,54-,55-,56-/m1/s1
WROHVVIPQXODQM-LYSKQWNXSA-N
Categorie correlate
Descrizione generale
Applicazioni
Carboxymethyl-β-cyclodextrin (CMBCD) is used in the development of recognition and separation technologies to resolve enantiomers based on chiral properties and size. Carboxymethyl-β-cyclodextrin is used in chiral selection and separation by capillary electrophoresis. Carboxymethyl-β-cyclodextrin is used in the development of drug delivery vehicles such as nanocarriers and as a nucleic acid transfection agent.
Risultati analitici
Altre note
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
Eyeshields, Gloves, type N95 (US)
Certificati d'analisi (COA)
Cerca il Certificati d'analisi (COA) digitando il numero di lotto/batch corrispondente. I numeri di lotto o di batch sono stampati sull'etichetta dei prodotti dopo la parola ‘Lotto’ o ‘Batch’.
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.