912654
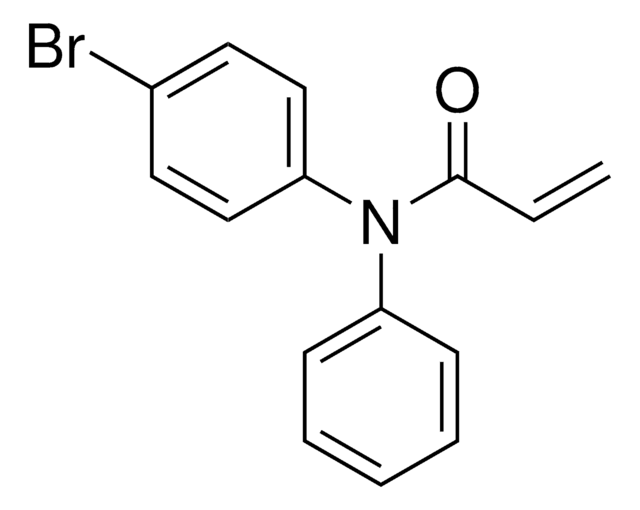
N-(3,5-Bis(trifluoromethyl)phenyl)-2-chloroacetamide
≥95%
Sinonimo/i:
N-Chloroacetyl-3,5-bis(trifluoromethyl)aniline, Electrophilic scout fragment, KB03, Scout fragment for targetable cysteine
About This Item
Prodotti consigliati
Saggio
≥95%
Stato
powder
InChI
1S/C10H6ClF6NO/c11-4-8(19)18-7-2-5(9(12,13)14)1-6(3-7)10(15,16)17/h1-3H,4H2,(H,18,19)
LEYIUTOAQOUAFG-UHFFFAOYSA-N
Applicazioni
Altre note
Note legali
Prodotti correlati
Avvertenze
Warning
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Eye Irrit. 2 - Skin Irrit. 2
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Scegli una delle versioni più recenti:
Certificati d'analisi (COA)
Non trovi la versione di tuo interesse?
Se hai bisogno di una versione specifica, puoi cercare il certificato tramite il numero di lotto.
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
Articoli
Ligandability describes the propensity of a protein target to bind a small molecule with high affinity. It is a precursor to evaluating druggability, which requires more advanced translational pharmacological effects and drug-like properties in vivo.
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 912654-100MG | 4061841784111 |
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.