905356
Rh2(R-PTAD)4
Sinonimo/i:
Tetrakis[(R)-(-)-(1-adamantyl)-(N-phthalimido)acetato]dirhodium(II)
About This Item
Prodotti consigliati
Stato
powder or crystals
Punto di fusione
>300 °C
Stringa SMILE
[Rh]([Rh](OC(=O)[C@H](N%19C(=O)c%20c(cccc%20)C%19=O)C%17%16C[C@@H]%18C[C@@H](C[C@@H](C%18)C%17)C%16)OC(=O)[C@H](N%14C(=O)c%15c(cccc%15)C%14=O)C%12%11C[C@@H]%13C[C@@H](C[C@@H](C%13)C%12)C%11)(OC(=O)[C@H](N9C(=O)c%10c(cccc%10)C9=O)C76C[C@@H]8C[C@@H](C[C@@H]
InChI
1S/4C20H21NO4.2Rh/c4*22-17-14-3-1-2-4-15(14)18(23)21(17)16(19(24)25)20-8-11-5-12(9-20)7-13(6-11)10-20;;/h4*1-4,11-13,16H,5-10H2,(H,24,25);;/q;;;;2*+2/p-4/t4*11-,12+,13-,16-,20?;;/m0000../s1
SGEDWOHAUXKUGM-VMLWFVNWSA-J
Applicazioni
Altre note
Asymmetric Synthesis of Tropanes by Rhodium-Catalyzed [4 + 3] Cycloaddition
Asymmetric [4 + 3] Cycloadditions between Vinylcarbenoids and Dienes: Application to the Total Synthesis of the Natural Product (−)-5-epi-Vibsanin E
Prodotti correlati
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Scegli una delle versioni più recenti:
Certificati d'analisi (COA)
It looks like we've run into a problem, but you can still download Certificates of Analysis from our Documenti section.
Se ti serve aiuto, non esitare a contattarci Servizio Clienti
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.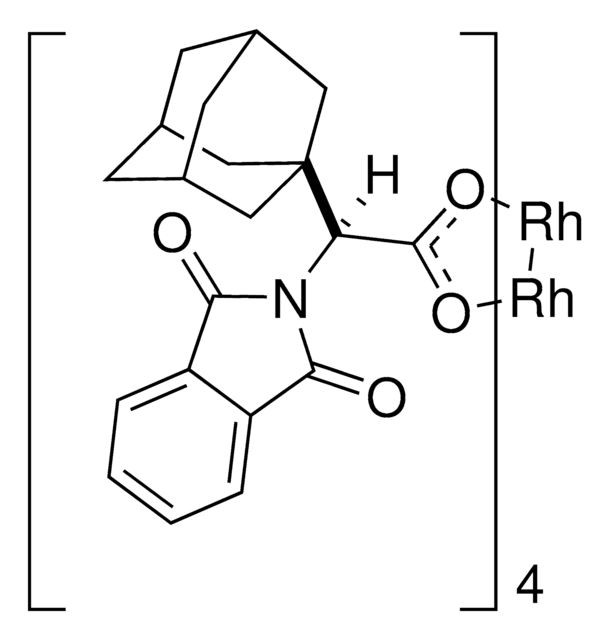

![Bis[rhodium(α,α,α′,α′-tetramethyl-1,3-benzenedipropionic acid)] 95%](/deepweb/assets/sigmaaldrich/product/structures/102/178/d1171a49-0358-406b-8b32-04324dbf9c02/640/d1171a49-0358-406b-8b32-04324dbf9c02.png)