512354
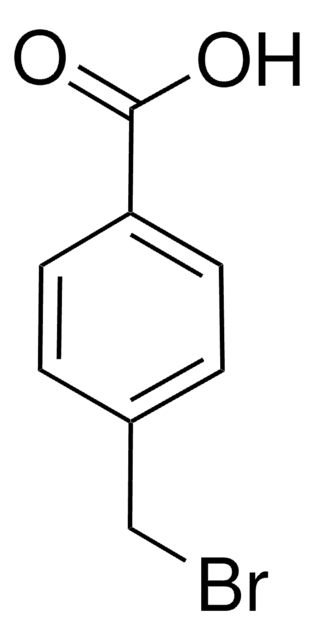
4-Benzyloxybenzoic acid
99%
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula condensata:
C6H5CH2OC6H4CO2H
Numero CAS:
Peso molecolare:
228.24
Numero CE:
Numero MDL:
Codice UNSPSC:
12352100
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Saggio
99%
Punto di fusione
189-192 °C (lit.)
Gruppo funzionale
carboxylic acid
phenyl
Stringa SMILE
OC(=O)c1ccc(OCc2ccccc2)cc1
InChI
1S/C14H12O3/c15-14(16)12-6-8-13(9-7-12)17-10-11-4-2-1-3-5-11/h1-9H,10H2,(H,15,16)
AQSCHALQLXXKKC-UHFFFAOYSA-N
Categorie correlate
Descrizione generale
4-Benzyloxybenzoic acid is a substituted benzoic acid that can be prepared by the benzylation of 4-hydroxybenzoic acid with benzyl bromide.
Applicazioni
4-Benzyloxybenzoic acid may be used in the preparation of 1,3-phenylene bis(4-benzyloxybenzoate). It may also be used in the preparation of (-)-(2R,3R)-5,7-bis(benzyloxy)-2-[3,4,5-tris(benzyloxy)-phenyl]chroman-3-yl-(4-benzyloxy)benzoate.
Avvertenze
Warning
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Acute Tox. 4 Oral - Eye Irrit. 2
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
dust mask type N95 (US), Eyeshields, Gloves
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
Synthesis and characterization of achiral banana-shaped liquid crystalline molecules containing bisnaphthyl moieties.
Yang PJ and Lin HC.
Liq. Cryst., 33(5), 587-603 (2006)
Henryk Marona et al.
Acta poloniae pharmaceutica, 60(6), 477-480 (2004-04-15)
A series of alkanolamides have been tested for anticonvulsant activity in the maximal electroshock seizure (MES) and subcutaneous pentylenetetrazole seizure treshold (ScMet) assays and for neurotoxicity (TOX) in rodents. Most interesting were the anticonvulsant results of 2N-methylaminoethanol derivative II, which
Kumi Osanai et al.
Tetrahedron, 63(32), 7565-7570 (2007-08-06)
The total and semi syntheses of (2R, 3R)-epigallocatechin-3-O-(4-hydroxybenzoate), a novel catechin from Cistus salvifolius, was accomplished. The proteasome inhibition and cytotoxic activities of the synthetic compound and its acetyl derivative were studied and compared with (2R, 3R)-epigallocatechin-3-gallate (EGCG), the active
Semi-synthesis and proteasome inhibition of D-ring deoxy analogs of (-)-epigallocatechin gallate (EGCG), the active ingredient of green tea extract.
Huo C, et al.
Canadian Journal of Chemistry, 86(6), 495-502 (2008)
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.