497312
4-Piperidinemethanol
97%
Sinonimo/i:
4-(Hydroxymethyl)piperidine
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula empirica (notazione di Hill):
C6H13NO
Numero CAS:
Peso molecolare:
115.17
Numero MDL:
Codice UNSPSC:
12352100
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Livello qualitativo
Saggio
97%
P. ebollizione
118-120 °C/10 mmHg (lit.)
Punto di fusione
55-59 °C (lit.)
Gruppo funzionale
hydroxyl
Stringa SMILE
OCC1CCNCC1
InChI
1S/C6H13NO/c8-5-6-1-3-7-4-2-6/h6-8H,1-5H2
XBXHCBLBYQEYTI-UHFFFAOYSA-N
Categorie correlate
Descrizione generale
4-Piperidinemethanol is a cyclic secondary amine. Its standard molar enthalpies of combustion, sublimation and formation have been determined.
Applicazioni
4-Piperidinemethanol may be used in the preparation of:
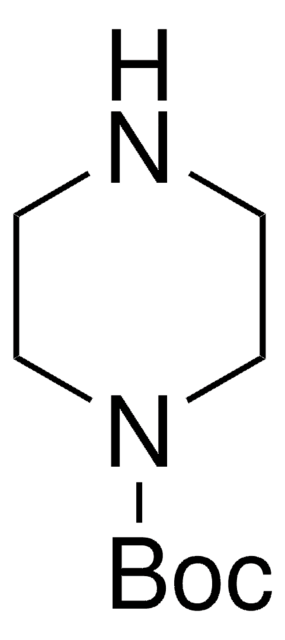
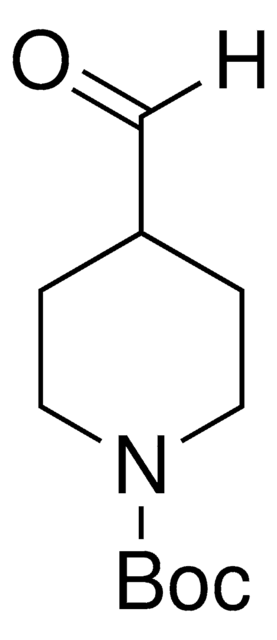
- N-tert-butoxycarbonyl-4-hydroxymethyl piperidine
- desferrioxamine B (DFO) containing third generation triazine dendrimer
- ethyl 3-(4-(hydroxymethyl)piperidin-1-yl)propanoate (EHMPP)
- 4-(hydroxymethyl)piperidine-1-carbodithioic acid (HL)
- 1-[[(1E)-2-(4-chlorophenyl)ethenyl]sulfonyl]-4-piperidinemethanol
Substrate used in solid-phase organic synthesis of a secondary amine.
Avvertenze
Danger
Indicazioni di pericolo
Classi di pericolo
Skin Corr. 1B
Codice della classe di stoccaggio
8A - Combustible corrosive hazardous materials
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
230.0 °F - closed cup
Punto d’infiammabilità (°C)
110 °C - closed cup
Dispositivi di protezione individuale
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Enzymatic synthesis, thermal and crystalline properties of a poly (β-amino ester) and poly (lactone-co-β-amino ester) copolymers.
Martino L, et al.
Polymer, 53(9), 1839-1848 (2012)
Synthesis and evaluation of 1-arylsulfonyl-3-piperazinone derivatives as factor Xa inhibitor.
Nishida H, et al.
Chemical & Pharmaceutical Bulletin, 49(10), 1237-1244 (2001)
Jongdoo Lim et al.
Bioorganic & medicinal chemistry, 18(15), 5749-5753 (2010-07-10)
The synthesis of a third generation triazine dendrimer, 1, containing multiple, iron-sequestering desferrioxamine B (DFO) groups is described. Benzoylation of the hydroxamic acid groups of DFO and formation of a reactive dichlorotriazine provide the intermediate for reaction with the second
Francis Giraud et al.
Bioorganic & medicinal chemistry letters, 19(2), 301-304 (2008-12-19)
Continuous efforts on the synthesis and structure-activity relationships (SARs) studies of modified 1-benzylamino-2-phenyl-3-(1H-1,2,4-triazol-1-yl)propan-2-ols as antifungal agents, allowed identification of new 1-[(pyridinyl- and piperidinylmethyl)amino] derivatives with MIC(80) values ranging from 1410.0 to 23.0ngmL(-1) on Candidaalbicans. These results confirmed both the importance
Synthesis, spectroscopy, and biological activity of heterobimetallic complexes containing Sn (IV) and Pd (II) with 4-(hydroxymethyl) piperidine-1-carbodithioic acid.
Anwar MT, et al.
Russ. J. Gen. Chem., 83(12), 2380-2385 (2013)
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.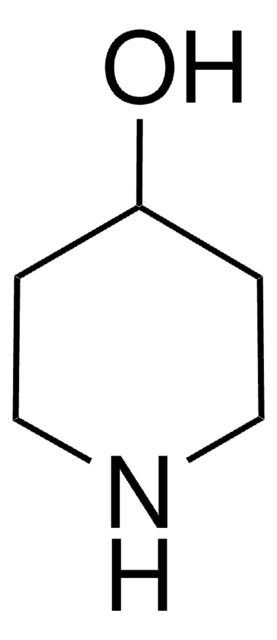
![[1,1′-Bis(diphenylphosphino)ferrocene]dichloropalladium(II)](/deepweb/assets/sigmaaldrich/product/structures/130/734/8846aa26-1858-458a-998d-8c306c13bf0f/640/8846aa26-1858-458a-998d-8c306c13bf0f.png)

![[1,1′-Bis(diphenylphosphino)ferrocene]dichloropalladium(II) complex with dichloromethane](/deepweb/assets/sigmaaldrich/product/structures/825/986/4317978b-1256-4c82-ab74-6a6a3ef948b1/640/4317978b-1256-4c82-ab74-6a6a3ef948b1.png)