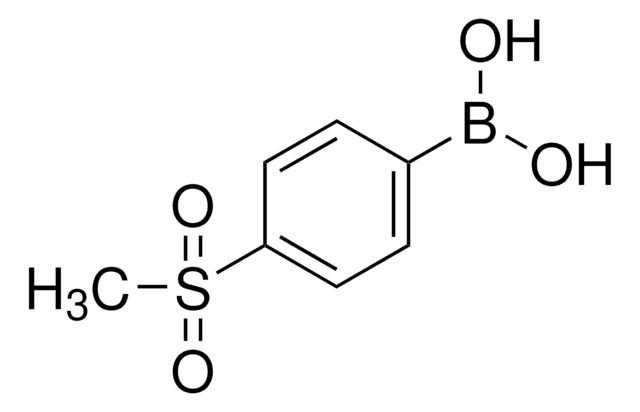
439320
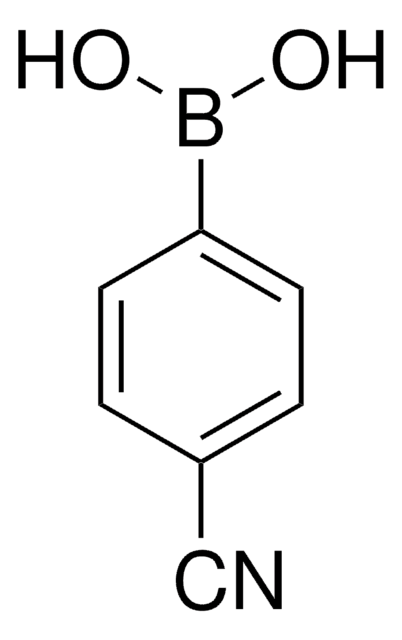
4-(Trifluoromethyl)phenylboronic acid
≥95.0%
Sinonimo/i:
α,α,α-Trifluoro-p-tolylboronic acid, 4-(Trifluoromethyl)benzeneboronic acid, [p-(Trifluoromethyl)phenyl]boronic acid
About This Item
Prodotti consigliati
Livello qualitativo
Saggio
≥95.0%
Punto di fusione
245-250 °C (lit.)
Gruppo funzionale
fluoro
Stringa SMILE
OB(O)c1ccc(cc1)C(F)(F)F
InChI
1S/C7H6BF3O2/c9-7(10,11)5-1-3-6(4-2-5)8(12)13/h1-4,12-13H
ALMFIOZYDASRRC-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Categorie correlate
Applicazioni
- Site-selective Suzuki-Miyaura cross-coupling reactions.
- Palladium-catalyzed direct arylation reactions.
- Tandem-type Pd(II)-catalyzed oxidative Heck reaction and intramolecular C-H amidation sequence.
- Ruthenium catalyzed direct arylation.
- Ligand-free copper-catalyzed coupling reactions.
- Amination and conjugate addition reactions.
- Regioselective arylation and alkynylation by Suzuki-Miyaura and Sonogashira cross-coupling reactions.
- Rhodium-catalyzed asymmetric 1,4-addition reactions.
- Copper-catalyzed nitration reactions.
- Regioselective Suzuki-Miyaura coupling and tandem palladium-catalyzed intramolecular aminocarbonylation and annulation.
- Palladium catalyzed allylation reaction with allyl alcohols.
- N-Arylation of imidazoles and amines in the presence of copper-exchanged fluorapatite as a catalyst.
It can also be used as a reactant to prepare:
- Thiazole derivatives for printable electronics.
- Terphenyl benzimidazoles as tubulin polymerization inhibitors.
- Aryl ketones by cross-coupling reaction with acid chlorides.
Altre note
Avvertenze
Warning
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Acute Tox. 4 Oral
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
dust mask type N95 (US), Eyeshields, Gloves
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.