279900
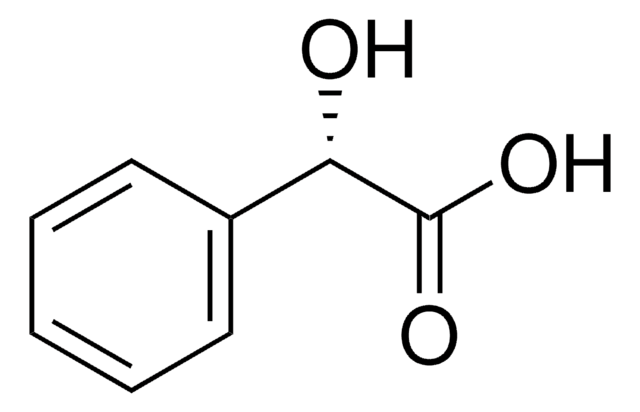
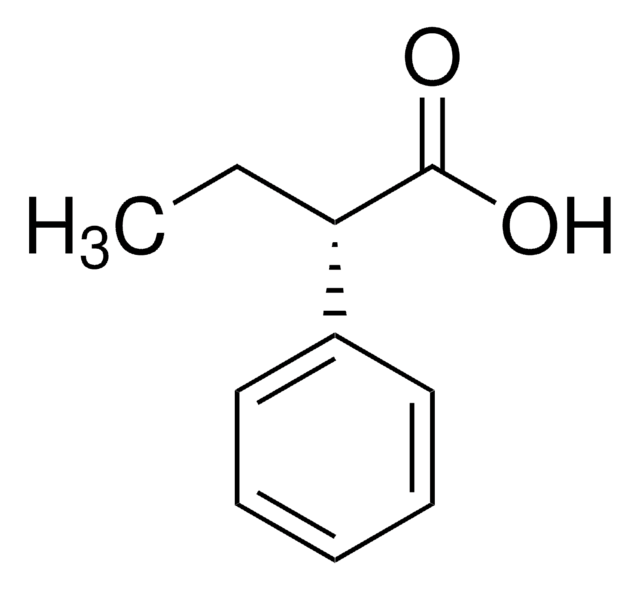
(S)-(+)-2-Phenylpropionic acid
97%
Sinonimo/i:
(S)-(+)-Hydratropic acid, (S)-HTA
About This Item
Prodotti consigliati
Saggio
97%
Stato
solid
Attività ottica
[α]20/D +72°, c = 1.6 in chloroform
Purezza ottica
ee: 98% (HPLC)
Indice di rifrazione
n20/D 1.522 (lit.)
P. ebollizione
115 °C/1 mmHg (lit.)
Punto di fusione
29-30 °C (lit.)
Densità
1.1 g/mL at 25 °C (lit.)
Gruppo funzionale
carboxylic acid
phenyl
Stringa SMILE
C[C@H](C(O)=O)c1ccccc1
InChI
1S/C9H10O2/c1-7(9(10)11)8-5-3-2-4-6-8/h2-7H,1H3,(H,10,11)/t7-/m0/s1
YPGCWEMNNLXISK-ZETCQYMHSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Applicazioni
Applicazioni
Avvertenze
Warning
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Eye Irrit. 2 - Skin Irrit. 2 - Skin Sens. 1 - STOT SE 3
Organi bersaglio
Respiratory system
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
>235.4 °F - closed cup
Punto d’infiammabilità (°C)
> 113 °C - closed cup
Dispositivi di protezione individuale
dust mask type N95 (US), Eyeshields, Faceshields, Gloves
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
Chromatograms
application for HPLCIl team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.