246352
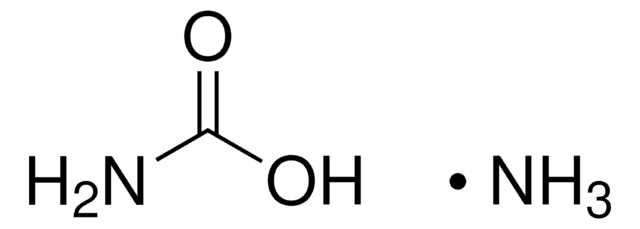
Methyl carbamate
98%
Sinonimo/i:
Urethylane
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula condensata:
NH2COOCH3
Numero CAS:
Peso molecolare:
75.07
Numero CE:
Numero MDL:
Codice UNSPSC:
12352100
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Livello qualitativo
Saggio
98%
Stato
crystals
P. ebollizione
176-177 °C (lit.)
Punto di fusione
56-58 °C (lit.)
Solubilità
alcohol: freely soluble(lit.)
water: freely soluble(lit.)
Gruppo funzionale
amine
Stringa SMILE
COC(N)=O
InChI
1S/C2H5NO2/c1-5-2(3)4/h1H3,(H2,3,4)
GTCAXTIRRLKXRU-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Applicazioni
Methyl carbamate was used in the synthesis of protected aminocyclopropanes.
Avvertenze
Warning
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Carc. 2 - Eye Irrit. 2
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Dispositivi di protezione individuale
Eyeshields, Gloves, type P3 (EN 143) respirator cartridges
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Shingo Ishikawa et al.
Angewandte Chemie (International ed. in English), 52(38), 10060-10063 (2013-08-06)
Easy as 1,2,3: Reaction of methyl carbamate, triethyl orthoformate, and readily available alkenes provides a highly practical preparation of protected aminocyclopropanes. The reaction proceeds with preferential cis addition to alkenes, and cleavage of the methyl carbamate gives the free aminocyclopropanes
John E Casida et al.
Chemico-biological interactions, 203(1), 221-225 (2012-08-29)
The anticholinesterase (antiChE) organophosphorus (OP) and methylcarbamate (MC) insecticides have been used very effectively as contact and systemic plant protectants for seven decades. About 90 of these compounds are still in use - the largest number for any insecticide chemotype
Xusheng Shao et al.
Proceedings of the National Academy of Sciences of the United States of America, 110(43), 17273-17277 (2013-10-11)
The nicotinic acetylcholine (ACh) receptor (nAChR) is the principal insecticide target. Nearly half of the insecticides by number and world market value are neonicotinoids acting as nAChR agonists or organophosphorus (OP) and methylcarbamate (MC) acetylcholinesterase (AChE) inhibitors. There was no
Catharina Wesseling et al.
Occupational and environmental medicine, 67(11), 778-784 (2010-08-28)
Neuropsychiatric disorders and increased suicide rates have been associated with exposure to cholinesterase inhibiting organophosphates. This study examined symptoms of psychological distress, including suicidal ideation, among banana workers in Costa Rica previously exposed to a cholinesterase inhibiting pesticide. 78 workers
P C Chan et al.
Japanese journal of cancer research : Gann, 83(3), 258-263 (1992-03-01)
Short-term and long-term carcinogenicity of methyl carbamate (MCB) was evaluated in F344 rats and B6C3F1 mice. In experiments lasting 6, 12, and 18 months, MCB was given in water by gavage to groups of 10 male and 10 female rats
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.