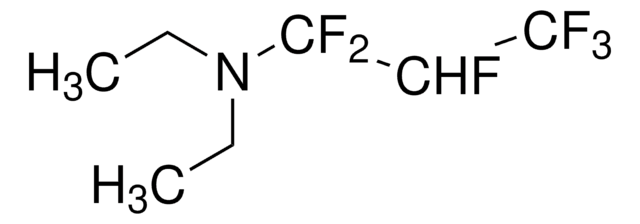
235253
(Diethylamino)sulfur trifluoride
95%
Sinonimo/i:
DAST, Diethylaminosulfur trifluoride
About This Item
Prodotti consigliati
Livello qualitativo
Saggio
95%
Stato
liquid
P. ebollizione
30-32 °C/3 mmHg (lit.)
Densità
1.22 g/mL at 25 °C (lit.)
Gruppo funzionale
amine
Temperatura di conservazione
2-8°C
Stringa SMILE
CCN(CC)S(F)(F)F
InChI
1S/C4H10F3NS/c1-3-8(4-2)9(5,6)7/h3-4H2,1-2H3
CSJLBAMHHLJAAS-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Categorie correlate
Descrizione generale
Applicazioni
- Fluorinating agent: reaction with alcohols and carbonyl compounds, Review
- Review on nucleophilic fluorination.
- Catalyst for Friedel-Crafts allylation using tertiary cyclopropyl silyl ethers and the rearrangement of homoallylic alcohols to unsaturated aldehydes.
- Early introduction of a fluoromethyl group stabilizes the epoxide during further manipulations in the synthesis of 26-fluoro-epothilone.
- Fluorinating agent for a variety of compounds, including thioethers, alkenols, and cyanohydrins.
- Reagent for gem difluorination of ketopipecolinic acids.
Sostituito da
Avvertenze
Danger
Indicazioni di pericolo
Classi di pericolo
Eye Dam. 1 - Flam. Liq. 3 - Self-react. D - Skin Corr. 1A
Rischi supp
Codice della classe di stoccaggio
5.2 - Organic peroxides and self-reacting hazardous materials
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
73.4 °F
Punto d’infiammabilità (°C)
23 °C
Dispositivi di protezione individuale
Faceshields, Gloves, Goggles, type ABEK (EN14387) respirator filter
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 235253-1G | 4061838784971 |
| 235253-5G | 4061838784995 |
| 235253-125G | 4061838231222 |
| 235253-250G | |
| 235253-25G | 4061838784988 |
| 235253-4X25G |
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.
![1-Chloromethyl-4-fluoro-1,4-diazoniabicyclo[2.2.2]octane bis(tetrafluoroborate) >95% in F+ active](/deepweb/assets/sigmaaldrich/product/structures/206/487/53d52ee5-ef71-4e9a-9bc8-938b68b98d5d/640/53d52ee5-ef71-4e9a-9bc8-938b68b98d5d.png)