228435
5-Bromopyridine-3-carboxylic acid
98%
Sinonimo/i:
5-Bromonicotinic acid
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula empirica (notazione di Hill):
C6H4BrNO2
Numero CAS:
Peso molecolare:
202.01
Beilstein:
115854
Numero CE:
Numero MDL:
Codice UNSPSC:
12352100
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Livello qualitativo
Saggio
98%
Forma fisica
solid
Punto di fusione
178-180 °C (lit.)
Gruppo funzionale
bromo
carboxylic acid
Stringa SMILE
OC(=O)c1cncc(Br)c1
InChI
1S/C6H4BrNO2/c7-5-1-4(6(9)10)2-8-3-5/h1-3H,(H,9,10)
FQIUCPGDKPXSLL-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Applicazioni
5-Bromopyridine-3-carboxylic acid (5-bromonicotinic acid) was used in the synthesis of 3-guanidinomethyl-5-iodopyridine.
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Dispositivi di protezione individuale
dust mask type N95 (US), Eyeshields, Gloves
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
G Vaidyanathan et al.
Bioconjugate chemistry, 9(6), 758-764 (1998-11-17)
Substituting a pyridine ring for a benzene ring in the acylation agent N-succinimidyl 3-iodobenzoate has resulted in a useful approach for the radiohalogenation of monoclonal antibodies, peptides, and labeled biotin conjugates. It was hypothesized that such a substitution in m-iodobenzylguanidine
Małgorzata Dukat et al.
European journal of pharmacology, 435(2-3), 171-180 (2002-02-01)
Two 5-substituted derivatives of nicotine (nicotinic acetylcholine receptor: K(i)=2.4 nM) were synthesized and evaluated: 5-bromonicotine (K(i)=6.9 nM) and 5-methoxynicotine (K(i)=14.3 nM). Despite their high affinity, neither 5-bromonicotine nor 5-methoxynicotine mimicked nicotine in producing antinociceptive (tail-flick, hotplate), hypolocomotor, or hypothermic effects
F Gabor et al.
Journal of pharmaceutical sciences, 84(9), 1120-1125 (1995-09-01)
Two types of monoclonal antibodies were used for the determination of nicergoline in biological matrices. The antibodies were prepared with the hydrolysis products 5-bromonicotinic acid and 1-methyl-10 alpha-methoxydihydrolysergol after hemisuccinoylation to haptens. The current amide bond-generating methods (mixed anhydride-, carbodiimide-
Tawfik Gharbaoui et al.
Bioorganic & medicinal chemistry letters, 17(17), 4914-4919 (2007-06-26)
A strategy for lead identification of new agonists of GPR109a, starting from known compounds shown to activate the receptor, is described. Early compound triage led to the formulation of a binding hypothesis and eventually to our focus on a series
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.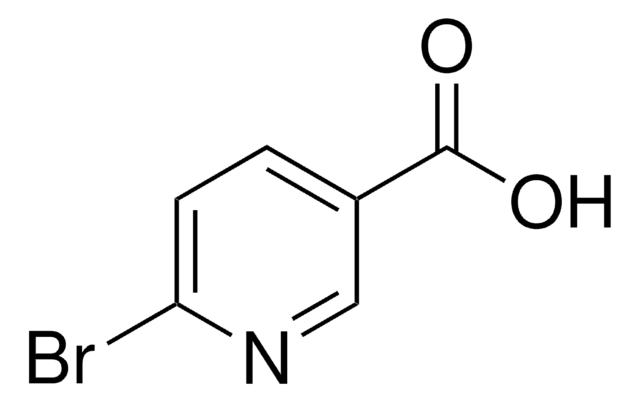




![[Pd(OAc)2]3 reagent grade, 98%](/deepweb/assets/sigmaaldrich/product/structures/508/249/99a0ef2c-b77c-4d73-8ed9-0cca05b6b41f/640/99a0ef2c-b77c-4d73-8ed9-0cca05b6b41f.png)
![[1,1′-Bis(diphenylphosphino)ferrocene]dichloropalladium(II)](/deepweb/assets/sigmaaldrich/product/structures/130/734/8846aa26-1858-458a-998d-8c306c13bf0f/640/8846aa26-1858-458a-998d-8c306c13bf0f.png)