193526
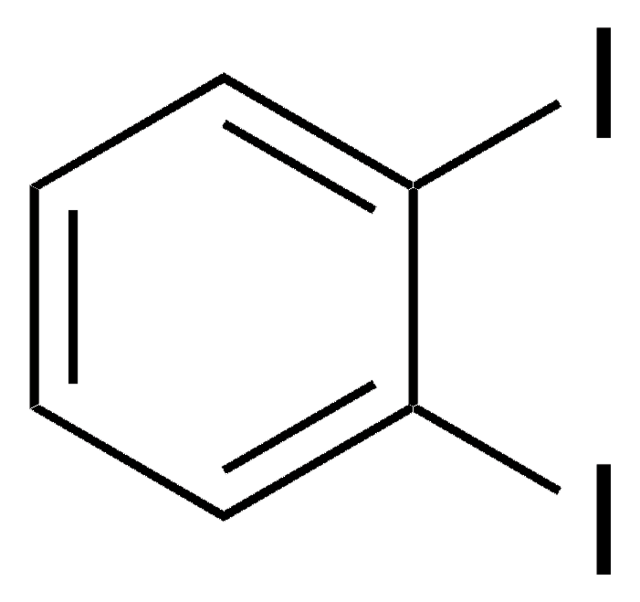
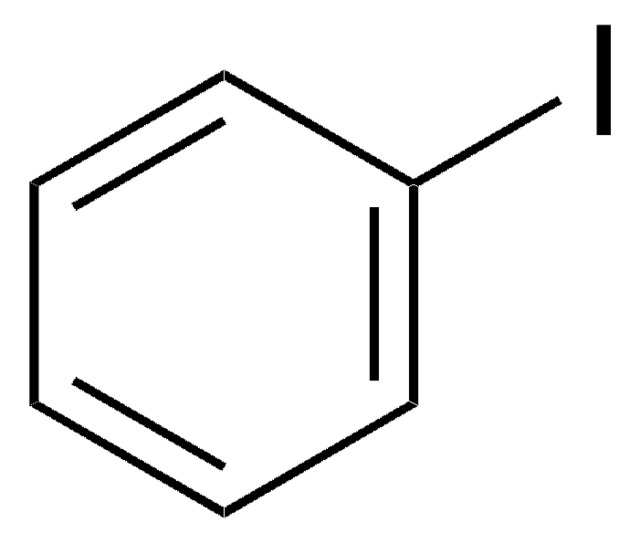
1,4-Diiodobenzene
99%
Sinonimo/i:
4-Iodophenyl iodide, p-Diiodobenzene, p-Phenylene diiodide
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula empirica (notazione di Hill):
C6H4I2
Numero CAS:
Peso molecolare:
329.90
Beilstein:
1904546
Numero CE:
Numero MDL:
Codice UNSPSC:
12352100
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Saggio
99%
Stato
powder
P. ebollizione
285 °C (lit.)
Punto di fusione
131-133 °C (lit.)
Gruppo funzionale
iodo
Stringa SMILE
Ic1ccc(I)cc1
InChI
1S/C6H4I2/c7-5-1-2-6(8)4-3-5/h1-4H
LFMWZTSOMGDDJU-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Categorie correlate
Applicazioni
1,4-Diiodobenzene was used in:
- total synthesis of martinellic acid, a naturally occurring bradykinin receptor antagonist
- preparation of 1,4-bis(p-R-phenylethynyl)benzenes via Pd11/Cu1catalyzed cross-coupling reaction
- synthesis of 1,4-diiodo-2,5-didodecylbenzene, starting reagent for the preparation of oligo(1,4-phenylene ethynylene)s
- surface-mediated synthesis of epitaxially aligned and separated polyphenylene lines on Cu(110) via Ullmann dehalogenation reaction
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
Eyeshields, Gloves, type N95 (US)
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Synthesis of symmetric and unsymmetric 1, 4-bis ( p-R-phenylethynyl) benzenes via palladium/copper catalyzed cross-coupling and comments on the coupling of aryl halides with terminal alkynes.
Nguyen P, et al.
Inorgorganica Chimica Acta, 220(1), 289-296 (1994)
D Ma et al.
Organic letters, 3(14), 2189-2191 (2001-07-07)
[reaction: see text] The first total synthesis of martinellic acid, a naturally occurring bradykinin receptor antagonist, via a CuI-catalyzed coupling reaction of beta-amino ester 6 with 1,4-diiodobenzene and a guanylation reaction of secondary amine 3 under mild conditions as key
Fabrication of a conjugated microporous polymer membrane and its application for membrane catalysis.
Jieun Lee et al.
Scientific reports, 7(1), 13568-13568 (2017-10-21)
A flexible and free standing conjugated microporous polymer (CMP) membrane was prepared using a polyvinylpyrrolidone (PVP) electrospun membrane as a template. The PVP nanofibers of the template membrane were coated with a thin layer of the CMP through the in
J A Lipton-Duffin et al.
Small (Weinheim an der Bergstrasse, Germany), 5(5), 592-597 (2009-02-26)
The surface-mediated synthesis of epitaxially aligned and separated polyphenylene lines on Cu(110) by exploiting the Ullmann dehalogenation reaction is reported. Scanning tunneling microscopy (STM) and X-ray photoelectron spectroscopy (XPS) show that the C-I bonds of 1,4-diiodobenzene and 1,3-diiodobenzene (C(6)H(4)I(2)) are
Uta Funke et al.
Pharmaceuticals (Basel, Switzerland), 5(2), 169-188 (2012-01-01)
Phosphodiesterase 10A (PDE10A) is a key enzyme of intracellular signal transduction which is involved in the regulation of neurotransmission. The molecular imaging of PDE10A by PET is expected to allow a better understanding of physiological and pathological processes related to
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.



![des-Arg9-[Leu8]-Bradykinin acetate salt ≥97% (HPLC)](/deepweb/assets/sigmaaldrich/product/structures/349/954/df5fe325-b9df-4769-80d8-10a3c754de46/640/df5fe325-b9df-4769-80d8-10a3c754de46.png)