136298
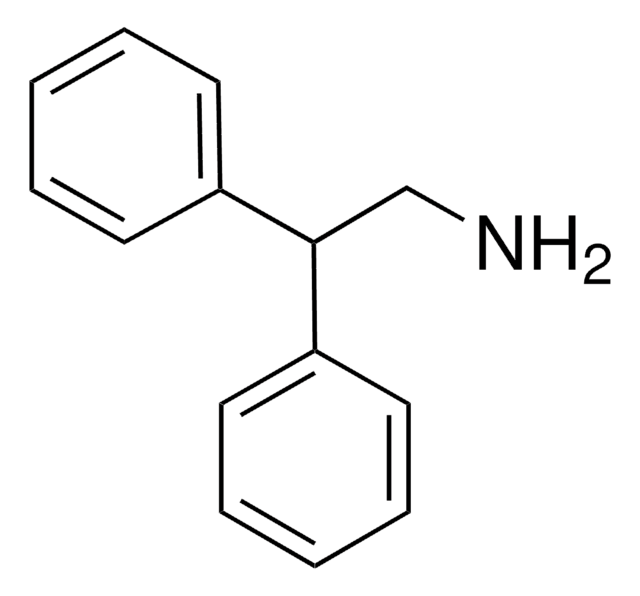
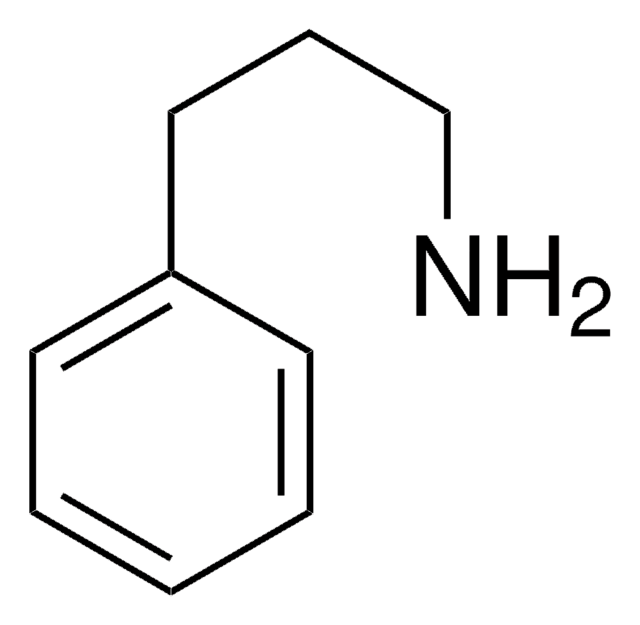
3,3-Diphenylpropylamine
97%
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
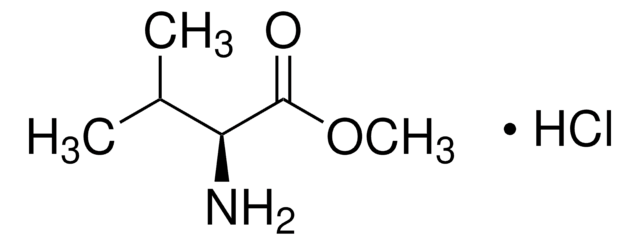
Formula condensata:
(C6H5)2CHCH2CH2NH2
Numero CAS:
Peso molecolare:
211.30
Numero CE:
Numero MDL:
Codice UNSPSC:
12352100
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Livello qualitativo
Saggio
97%
Indice di rifrazione
n20/D 1.583 (lit.)
Punto di fusione
29-31 °C (lit.)
Gruppo funzionale
amine
phenyl
Stringa SMILE
NCCC(c1ccccc1)c2ccccc2
InChI
1S/C15H17N/c16-12-11-15(13-7-3-1-4-8-13)14-9-5-2-6-10-14/h1-10,15H,11-12,16H2
KISZTEOELCMZPY-UHFFFAOYSA-N
Applicazioni
3,3-Diphenylpropylamine was used as internal standard for simultaneous determination of D- and L-modafinil in human plasma using stereospecific high-performance liquid chromatographic method. It was used as starting reagent in the synthesis of 3,3-diphenylpropylisocyanate.
Avvertenze
Warning
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Organi bersaglio
Respiratory system
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
235.4 °F - closed cup
Punto d’infiammabilità (°C)
113 °C - closed cup
Dispositivi di protezione individuale
dust mask type N95 (US), Eyeshields, Gloves
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
A L Mueller et al.
Annals of the New York Academy of Sciences, 890, 450-457 (2000-02-11)
NPS 1506 is a moderate affinity, uncompetitive N-methyl-D-aspartate (NMDA) receptor antagonist. NPS 1506 is neuroprotective in rodent models of ischemic stroke, hemorrhagic stroke, and head trauma, with a 2-hr window of opportunity. Neuroprotectant doses of NPS 1506 ranged from approximately
B Elsenhans et al.
Biochimica et biophysica acta, 813(1), 25-32 (1985-02-28)
Cationic, lipid-soluble organic compounds may interfere with cation-mediated membrane transport processes. Thus, small intestinal absorption may be influenced by lipophilic organic cations. Therefore a series of arylalkylamines was studied in the concentration range from 0.5 to 20 mmol/l for their
S H Gorman
Journal of chromatography. B, Biomedical sciences and applications, 730(1), 1-7 (1999-08-07)
Modafinil, DL-2-[(diphenylmethyl)sulfinyl]acetamide (Provigil), which is chiral at its sulfur atom, is a novel wake-promoting agent currently being developed as the racemate in the United States by Cephalon, Inc. In order to characterize the pharmacokinetic properties of each enantiomer, a stereospecific
T F Holzman et al.
Biochemistry, 20(19), 5524-5528 (1981-09-15)
We have found a new class of inhibitors of the bacterial bioluminescence reaction, the N,N-diphenylalkylamines and acids. We have studied the action of one of these compounds 2,2-diphenylpropylamine. The amine was competitive with the long-chain aliphatic aldehyde substrate (Ki congruent
Shiv K Sharma et al.
Journal of medicinal chemistry, 53(14), 5197-5212 (2010-06-24)
The recently discovered enzyme lysine-specific demethylase 1 (LSD1) plays an important role in the epigenetic control of gene expression, and aberrant gene silencing secondary to LSD1 overexpression is thought to contribute to the development of cancer. We recently reported a
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.