G3798
10074-G5
≥98% (HPLC)
别名:
Biphenyl-2-yl-(7-nitrobenzo[1,2,5]oxadiazol-4-yl)amine, N-2-Biphenylyl-7-nitro-2,1,3-benzoxadiazol-4-amine, N-[1,1′-Biphenyl-2-yl]-7-nitro-2,1,3-Benzoxadiazol-4-amine
登录查看公司和协议定价
所有图片(1)
About This Item
经验公式(希尔记法):
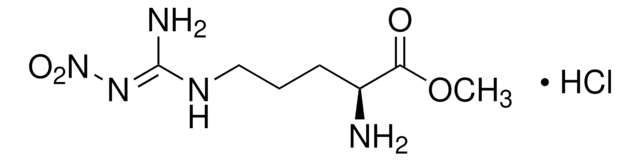
C18H12N4O3
CAS号:
分子量:
332.31
MDL號碼:
分類程式碼代碼:
12352200
PubChem物質ID:
NACRES:
NA.77
推荐产品
品質等級
化驗
≥98% (HPLC)
形狀
powder
顏色
red
溶解度
DMSO: >10 mg/mL
儲存溫度
room temp
SMILES 字串
[O-][N+](=O)c1ccc(Nc2ccccc2-c3ccccc3)c4nonc14
InChI
1S/C18H12N4O3/c23-22(24)16-11-10-15(17-18(16)21-25-20-17)19-14-9-5-4-8-13(14)12-6-2-1-3-7-12/h1-11,19H
InChI 密鑰
KMJPYSQOCBYMCF-UHFFFAOYSA-N
生化/生理作用
10074-G5 is a c-Myc/Max interaction inhibitor. The c-Myc oncoprotein and its partner Max are intrinsically disordered (ID) monomers that undergo coupled folding and binding upon heterodimerization. 10074-G5, similarly to 10058-F4 (#F3680), specifically inhibits this interaction by binding to c-Myc, thus preventing C-Myc specific DNA binding and target genes regulation. 10074-G5 (2.8 microM) is slightly more potent that 10058-F4 (5.2 microM). It was discovered that 10074-G5 binds to a different specific binding site (region) of C-Myc than 10054-F4. Thus, the compound may become desirable for probing different interactions.
儲存類別代碼
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
水污染物質分類(WGK)
WGK 3
Chengsheng Wu et al.
BMC cancer, 18(1), 361-361 (2018-04-04)
The phenomenon that malignant cells can acquire stemness under specific stimuli, encompassed under the concept of cancer cell plasticity, has been well-described in epithelial malignancies. To our knowledge, cancer cell plasticity has not yet been described in hematopoietic cancers. To
Quan Yang et al.
Frontiers in immunology, 12, 627072-627072 (2021-03-13)
The accumulation of myeloid-derived suppressor cells (MDSCs) is one of the major obstacles to achieve an appropriate anti-tumor immune response and successful tumor immunotherapy. MDSCs in tumor-bearing hosts are primarily polymorphonuclear (PMN-MDSCs). However, the mechanisms regulating the development of MDSCs
Alina Castell et al.
Scientific reports, 8(1), 10064-10064 (2018-07-04)
MYC is a key player in tumor development, but unfortunately no specific MYC-targeting drugs are clinically available. MYC is strictly dependent on heterodimerization with MAX for transcription activation. Aiming at targeting this interaction, we identified MYCMI-6 in a cell-based protein
Udom Lao-On et al.
Biochimica et biophysica acta. Molecular basis of disease, 1866(3), 165656-165656 (2019-12-25)
Here we showed that the c-Myc oncogene is responsible for overexpression of pyruvate carboxylase (PC) in highly invasive MDA-MB-231 cells. Pharmacological inhibition of c-Myc activity with 10074-G5 compound, resulted in a marked reduction of PC mRNA and protein, concomitant with
Huabo Wang et al.
Oncotarget, 6(18), 15857-15870 (2015-06-04)
The c-Myc (Myc) oncoprotein is deregulated in a large proportion of diverse human cancers. Considerable effort has therefore been directed at identifying pharmacologic inhibitors as potential anti-neoplastic agents. Three such groups of small molecule inhibitors have been described. The first
商品
We present an article about how proliferating cells require the biosynthesis of structural components for biomass production and for genomic replication.
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门