SML0593
CGP52432
≥98% (HPLC)
Sinónimos:
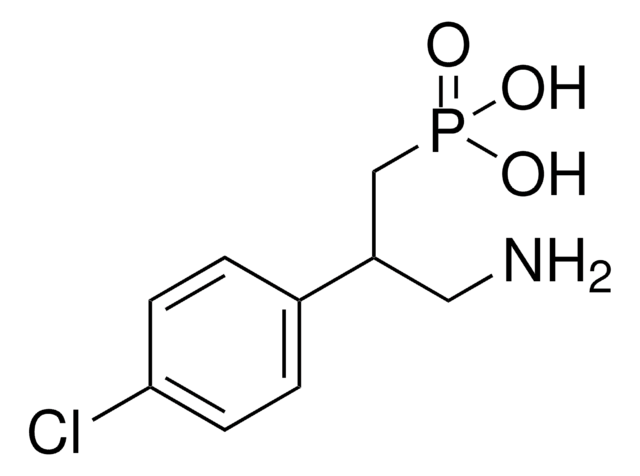
[3-[[(3,4-Dichlorophenyl)methyl]amino]propyl](diethoxymethyl)-phosphinic acid
About This Item
Productos recomendados
Análisis
≥98% (HPLC)
formulario
powder
condiciones de almacenamiento
desiccated
color
white to beige
solubilidad
H2O: 1 mg/mL, clear (warmed)
temp. de almacenamiento
2-8°C
InChI
1S/C15H24Cl2NO4P/c1-3-21-15(22-4-2)23(19,20)9-5-8-18-11-12-6-7-13(16)14(17)10-12/h6-7,10,15,18H,3-5,8-9,11H2,1-2H3,(H,19,20)
Clave InChI
GJZVQXWEIYRHBE-UHFFFAOYSA-N
Aplicación
- to study its effects on the simulation of the onset of status epilepticus (SE) in mice
- for voltage-clamp recording in mice neurons
- to study its effects on the GABAB receptor-mediated neurotransmission in guinea pig hippocampus
Acciones bioquímicas o fisiológicas
Características y beneficios
Otras notas
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico