04991
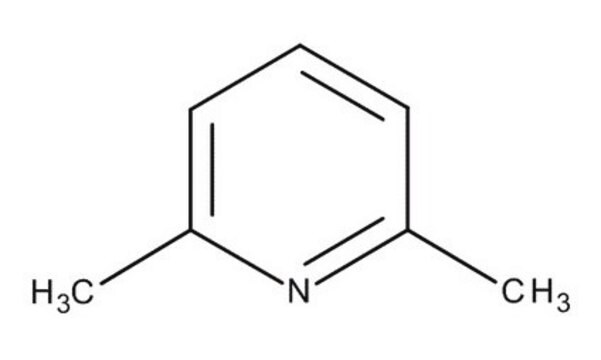
2,6-Lutidina
analytical standard
Sinónimos:
2,6-Dimetilpiridina
About This Item
Productos recomendados
grado
analytical standard
Nivel de calidad
Ensayo
≥99.7% (GC)
caducidad
limited shelf life, expiry date on the label
técnicas
HPLC: suitable
gas chromatography (GC): suitable
índice de refracción
n20/D 1.497 (lit.)
n20/D 1.499
bp
143-145 °C (lit.)
mp
−6 °C (lit.)
densidad
0.92 g/mL at 25 °C (lit.)
aplicaciones
cleaning products
cosmetics
flavors and fragrances
food and beverages
personal care
Formato
neat
cadena SMILES
Cc1cccc(C)n1
InChI
1S/C7H9N/c1-6-4-3-5-7(2)8-6/h3-5H,1-2H3
Clave InChI
OISVCGZHLKNMSJ-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Palabra de señalización
Warning
Frases de peligro
Consejos de prudencia
Clasificaciones de peligro
Acute Tox. 4 Oral - Eye Irrit. 2 - Flam. Liq. 3 - Skin Irrit. 2
Código de clase de almacenamiento
3 - Flammable liquids
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
89.6 °F
Punto de inflamabilidad (°C)
32 °C
Equipo de protección personal
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
Elija entre una de las versiones más recientes:
Certificados de análisis (COA)
¿No ve la versión correcta?
Si necesita una versión concreta, puede buscar un certificado específico por el número de lote.
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico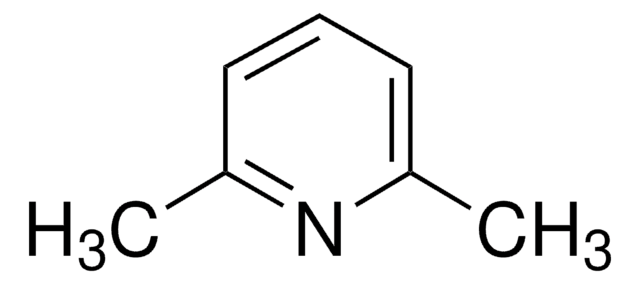


![1,8-Diazabiciclo[5.4.0]undec-7-eno 98%](/deepweb/assets/sigmaaldrich/product/structures/120/564/5b373e23-1624-489c-8efb-692de0f96ffb/640/5b373e23-1624-489c-8efb-692de0f96ffb.png)