W323713
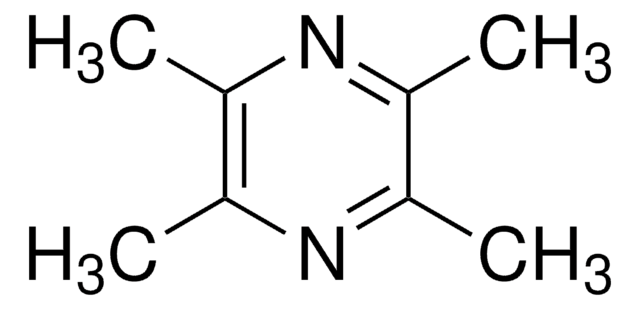
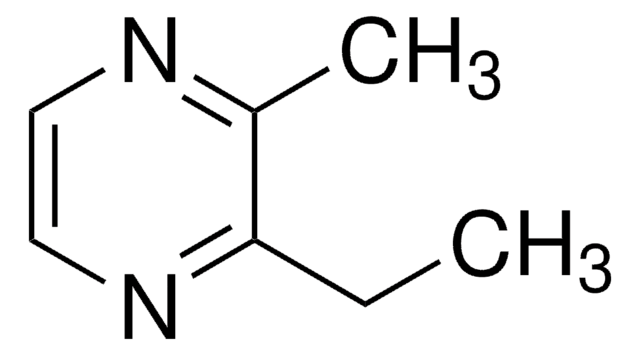
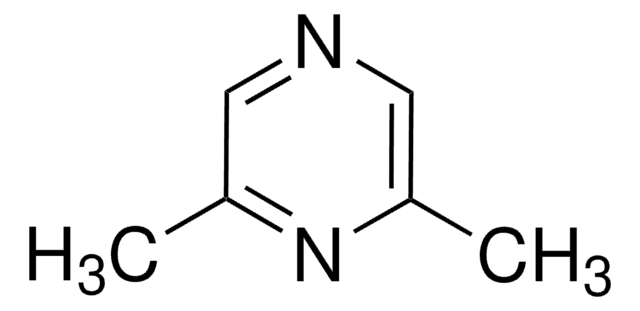
2,3,5,6-Tetramethylpyrazine
natural, ≥98%, FG
Sinónimos:
Chuanxingzine, Ligustrazine, Tetrapyrazine
About This Item
Productos recomendados
grado
FG
Fragrance grade
Halal
Kosher
natural
Nivel de calidad
Agency
follows IFRA guidelines
meets purity specifications of JECFA
cumplimiento norm.
EU Regulation 1223/2009
EU Regulation 1334/2008 & 178/2002
Ensayo
≥98%
características de los productos alternativos más sostenibles
Less Hazardous Chemical Syntheses
Use of Renewable Feedstocks
Learn more about the Principles of Green Chemistry.
sustainability
Greener Alternative Product
impurezas
≤2.0% water (Karl Fischer)
bp
190 °C (lit.)
mp
77-80 °C (lit.)
aplicaciones
flavors and fragrances
Documentación
see Safety & Documentation for available documents
alérgeno alimentario
no known allergens
alérgeno de la fragancia
no known allergens
categoría alternativa más sostenible
, Aligned
Organoléptico
chocolate; coffee; fatty; musty; nutty
cadena SMILES
Cc1nc(C)c(C)nc1C
InChI
1S/C8H12N2/c1-5-6(2)10-8(4)7(3)9-5/h1-4H3
Clave InChI
FINHMKGKINIASC-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Descripción general
Aplicación
- Booklice Liposcelis bostrychophila are efficiently attracted by the combination of 2,3,5,6-tetramethylpyrazine and ultraviolet light.: This study demonstrates that the combination of 2,3,5,6-tetramethylpyrazine and ultraviolet light effectively attracts booklice, suggesting a potential application for pest management in stored product environments (Tanaka et al., 2024).
- 2,3,5,6-Tetramethylpyrazine protects retinal photoreceptors against endoplasmic reticulum stress by modulating ATF4-mediated inhibition of PRP aggregation.: The research highlights the neuroprotective effects of 2,3,5,6-tetramethylpyrazine, showing its potential in treating retinal diseases by protecting photoreceptors from stress-induced damage (Huang et al., 2021).
- Tetramethylpyrazine-Inducible Promoter Region from Rhodococcus jostii TMP1.: The study identifies a promoter region in Rhodococcus jostii TMP1 that is inducible by tetramethylpyrazine, which could be utilized in genetic engineering and biotechnology applications (Stanislauskienė et al., 2018).
Palabra de señalización
Warning
Frases de peligro
Consejos de prudencia
Clasificaciones de peligro
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Órganos de actuación
Respiratory system
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Equipo de protección personal
dust mask type N95 (US), Eyeshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico