433063
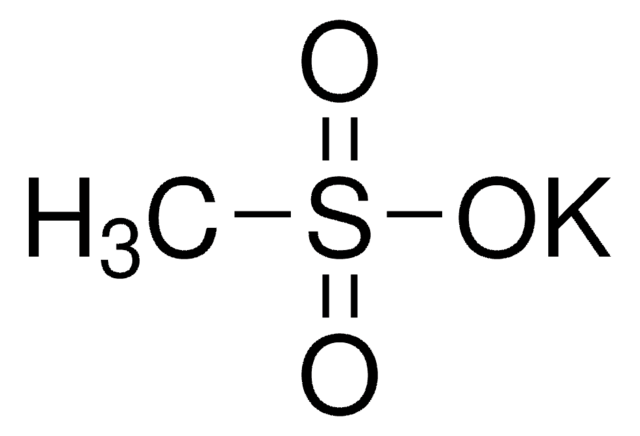
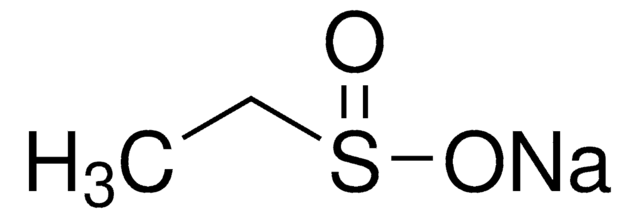
Sodium methanesulfinate
technical grade, 85%
Sinónimos:
Methanesulfinic acid sodium salt
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
Fórmula lineal:
CH3SO2Na
Número de CAS:
Peso molecular:
102.09
Número CE:
Número MDL:
Código UNSPSC:
12352100
ID de la sustancia en PubChem:
NACRES:
NA.22
Productos recomendados
grado
technical grade
Ensayo
85%
mp
222-226 °C (dec.) (lit.)
grupo funcional
sulfinic acid
cadena SMILES
[Na+].CS([O-])=O
InChI
1S/CH4O2S.Na/c1-4(2)3;/h1H3,(H,2,3);/q;+1/p-1
Clave InChI
LYPGDCWPTHTUDO-UHFFFAOYSA-M
¿Está buscando productos similares? Visita Guía de comparación de productos
Descripción general
Sodium methanesulfinate is an aliphatic sodium sulfinate. Conjugate addition of sodium methanesulfinate to vinyl heterocycles has been described. Cross-coupling reaction between aryl boronic acid and sodium methanesulfinate has been studied. Its stock solution was prepared from methanesulfonic acid by adding one equivalent of sodium hydroxide and diluting it to 4M.
Palabra de señalización
Warning
Frases de peligro
Clasificaciones de peligro
Acute Tox. 4 Oral
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Equipo de protección personal
dust mask type N95 (US), Eyeshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Metal-Free Direct Construction of Sulfonamides via Iodine-Mediated Coupling Reaction of Sodium Sulfinates and Amines at Room Temperature.
Wei W, et al.
Advanced Synthesis & Catalysis, 357(5), 987-992 (2015)
J B Smith et al.
Free radical research communications, 8(2), 101-106 (1990-01-01)
Iron bound to certain chelators is known to promote the conversion of superoxide radicals (O2-) to hydroxyl radicals (HO.) by the superoxide-driven Fenton reaction. The production of HO. by various iron chelates was studied using the reaction of dimethyl sulfoxide
M G Steiner et al.
Archives of biochemistry and biophysics, 278(2), 478-481 (1990-05-01)
This investigation was conducted to validate the use of dimethyl sulfoxide (DMSO) as a quantitative molecular probe for the generation of hydroxyl radicals (HO.) in aqueous systems. Reaction of HO. with DMSO produces methane sulfinic acid as a primary product
M G Steiner et al.
Free radical biology & medicine, 9(1), 67-77 (1990-01-01)
To quantitate the formation of hydroxyl radicals (HO.) in ischemia and reoxygenation, dimethyl sulfoxide (DMSO) was added to "trap" evolving HO. in normal, in ischemic, and in ischemic and reoxygenated rat kidney slices, incubated in short-term organ culture in vitro.
S Fukui et al.
Journal of chromatography, 630(1-2), 187-193 (1993-02-05)
For the determination of hydroxyl radicals, dimethyl sulphoxide was used as a molecular probe and the methanesulphinic acid produced was determined by high-performance liquid chromatography of its Fast Yellow GC salt derivative. The results for hydroxyl radicals formed using the
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico

![1,8-Diazabiciclo[5.4.0]undec-7-eno 98%](/deepweb/assets/sigmaaldrich/product/structures/120/564/5b373e23-1624-489c-8efb-692de0f96ffb/640/5b373e23-1624-489c-8efb-692de0f96ffb.png)