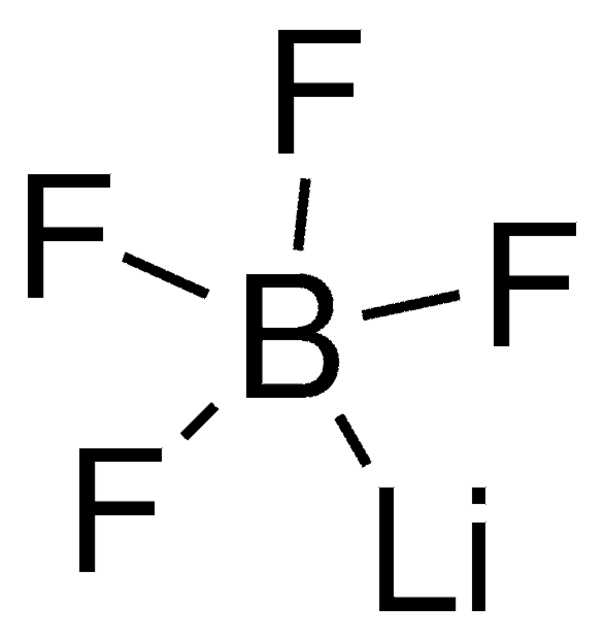
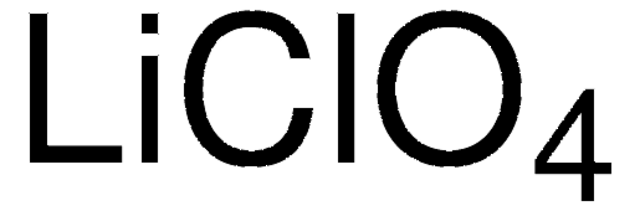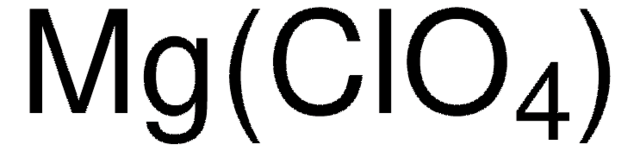431567
Lithium perchlorate
99.99% trace metals basis
Sinónimos:
Perchloric acid, lithium salt
About This Item
Productos recomendados
grado
ACS reagent
Nivel de calidad
Ensayo
99.99% trace metals basis
Formulario
granular
idoneidad de la reacción
reagent type: oxidant
características de los productos alternativos más sostenibles
Design for Energy Efficiency
Learn more about the Principles of Green Chemistry.
sustainability
Greener Alternative Product
impurezas
≤0.005% insolubles
<100 ppm total metallic impurities
pH
6.0-7.5
mp
236 °C (lit.)
solubilidad
H2O: 106.4 g/L at 20 °C
trazas de anión
chloride (Cl-): ≤0.003%
sulfate (SO42-): ≤0.001%
categoría alternativa más sostenible
cadena SMILES
[Li+].[O-]Cl(=O)(=O)=O
InChI
1S/ClHO4.Li/c2-1(3,4)5;/h(H,2,3,4,5);/q;+1/p-1
Clave InChI
MHCFAGZWMAWTNR-UHFFFAOYSA-M
¿Está buscando productos similares? Visita Guía de comparación de productos
Categorías relacionadas
Descripción general
Aplicación
- As a precursor to prepare solid polymer electrolytes for rechargeable Li-ion batteries.
- As an oxidizer to prepare polymer-based solid propellants.
- To fabricate cobalt sulfide (CoS)-based counter electrode for dye-sensitized solar cells(DSSC).
Palabra de señalización
Danger
Frases de peligro
Consejos de prudencia
Clasificaciones de peligro
Acute Tox. 4 Oral - Eye Dam. 1 - Ox. Sol. 2 - Skin Corr. 1A - STOT SE 3
Órganos de actuación
Respiratory system
Código de clase de almacenamiento
5.1A - Strongly oxidizing hazardous materials
Clase de riesgo para el agua (WGK)
WGK 1
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Equipo de protección personal
Eyeshields, Gloves, type P3 (EN 143) respirator cartridges
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Global Trade Item Number
| Número de referencia del producto (SKU) | GTIN |
|---|---|
| 431567-250G | 4061832108346 |
| 431567-50G | 4061832108353 |
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico