275603
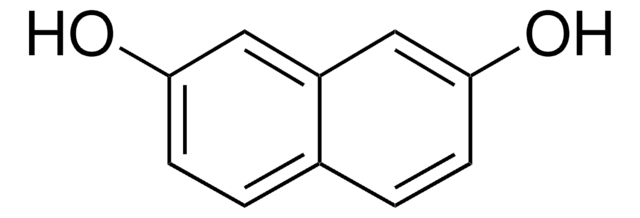
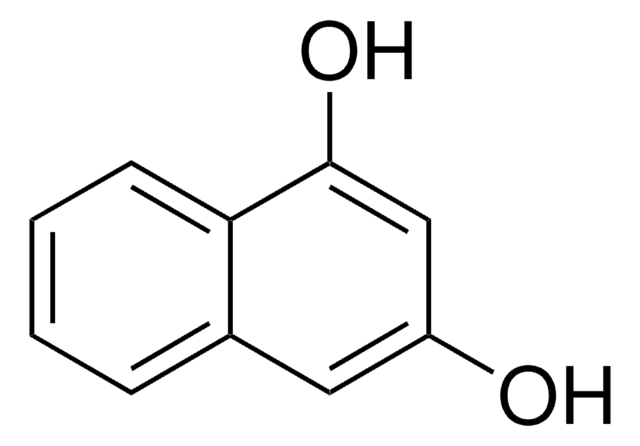
2,6-Dihydroxynaphthalene
98%
Sinónimos:
2,6-Naphthalenediol
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
Fórmula lineal:
C10H6(OH)2
Número de CAS:
Peso molecular:
160.17
Beilstein:
1238082
Número CE:
Número MDL:
Código UNSPSC:
12352100
ID de la sustancia en PubChem:
NACRES:
NA.22
Productos recomendados
Análisis
98%
formulario
solid
mp
223-225 °C (lit.)
cadena SMILES
Oc1ccc2cc(O)ccc2c1
InChI
1S/C10H8O2/c11-9-3-1-7-5-10(12)4-2-8(7)6-9/h1-6,11-12H
Clave InChI
MNZMMCVIXORAQL-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Aplicación
2,6-Dihydroxynaphthalene was used in the synthesis of 1,5-dichloro-2,6-diethynylnaphthalenes. It was also used in the preparation of first-generation rotaxane dendrimer.
Palabra de señalización
Warning
Frases de peligro
Consejos de prudencia
Clasificaciones de peligro
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Órganos de actuación
Respiratory system
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 2
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Equipo de protección personal
dust mask type N95 (US), Eyeshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Shoji Shinamura et al.
The Journal of organic chemistry, 75(4), 1228-1234 (2010-01-27)
In this paper we present the synthesis, structures, characterization, and applications to field-effect transistors (FETs) of naphtho[1,2-b:5,6-b']dithiophene (NDT) and -diselenophene (NDS) derivatives. Treatment of 1,5-dichloro-2,6-diethynylnaphthalenes, easily derived from commercially available 2,6-dihydroxynaphthalene, with sodium chalcogenide afforded a straightforward access to NDTs
Soo-Young Kim et al.
Chemistry, an Asian journal, 2(6), 747-754 (2007-05-08)
By taking advantage of the fact that cucurbit[6]uril (CB[6]) forms exceptionally stable host-guest complexes with protonated amines, and that its homologue CB[8] can encapsulate a pair of electron-rich and electron-deficient guest molecules to form a stable 1:1:1 complex, we synthesized
Alberto Macone et al.
Bioorganic & medicinal chemistry, 17(16), 6003-6007 (2009-07-21)
Aromatic substrates tyrosol (p-hydroxyphenylethanol) and 2,6-dihydroxynaphthalene (2,6-DHN) were converted into chromane derivatives by means of chemoenzymatic reactions catalyzed by the aromatic prenyltransferase of bacterial origin NovQ, using dimethylallyl bromide as allylic substrate instead of the natural isoprenyl pyrophosphate substrate. Stereoselective
Mårten Jacobsson et al.
Journal of medicinal chemistry, 49(6), 1932-1938 (2006-03-17)
The antiproliferative activity of the 14 isomeric monoxylosylated dihydroxynaphthalenes has been tested in vitro toward normal HFL-1 and 3T3 A31 cells as well as transformed T24 and 3T3 SV40 cells. The antiproliferative effect toward HFL-1 cells was correlated with the
Dieter Schemeth et al.
Analytica chimica acta, 1038, 182-190 (2018-10-04)
In this study, we focus on isolation and fractionation strategies by solid phase extraction (SPE) for a broad range of environmentally related organic acids. These emerging potential contaminants are primary degradation products of spilled petrogenic compounds but little attention has
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico