TZHVCA210
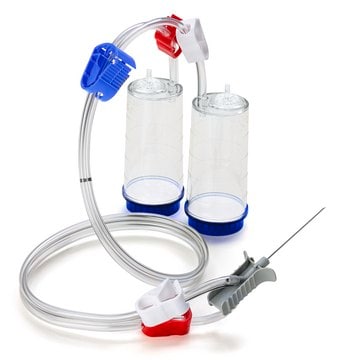
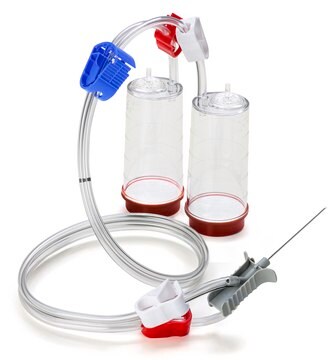
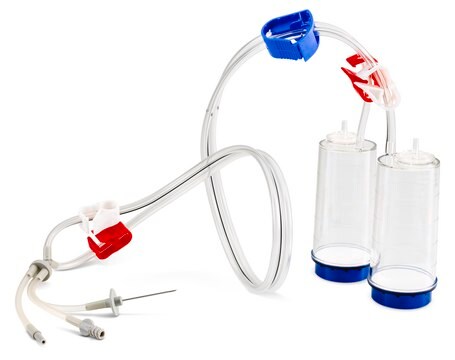
Steritest® NEO Device
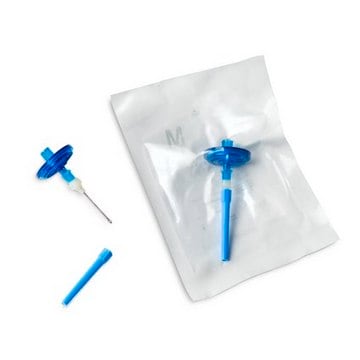
For liquids in cartridges and small soft plastic containers. Red base canister comes with a single short (20 mm) needle.
Synonym(e):
Red Base Steritest® NEO device for sterility testing, Sterility testing device, membrane filtration device, membrane filtration canister, closed membrane filtration
About This Item
Empfohlene Produkte
Materialien
Nylon 66 adapter (for needle)
PVC tubing (double lumen)
PVDF membrane
stainless steel (for needle)
styrene-acrylonitrile (SAN) (for canister)
Agentur
EP 2.6.1
JP 4.06
USP 71
Sterilität
sterile; γ-irradiated
Hersteller/Markenname
Steritest®
Verpackung
pkg of 10 blisters per box (Single packed)
Parameter
120 mL sample volume (graduation marks at 25, 50, 75 and 100 mL)
3.1 bar max. inlet pressure (45 psi) at 25 °C
45 °C max. temp.
Länge Schlauch
850 mm
Farbe
red Canister Base
Matrix
Durapore®
Porengröße
0.45 μm pore size
Aufnahme
liquid
pharmaceutical(s)
Anwendung(en)
pharmaceutical
sterility testing
Kompatibilität
for use with Steritest® Symbio FLEX Pump Kit, 2 media (SYMBFLE01)
for use with Steritest® Symbio ISL Pump Kit, 2 media (SYMBISL01)
for use with Steritest® Symbio LFH Pump Kit (SYMBLFH01)
Allgemeine Beschreibung
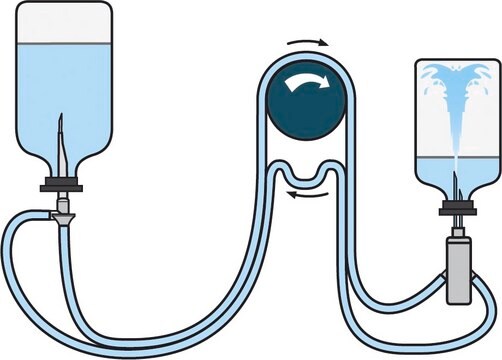
Steritest® NEO is a membrane filtration device for sterility testing of filterable pharmaceutical products. The device simplifies every aspect of testing, from handling to traceability. The closed system minimizes false positives and offers the highest levels of quality and reliability. This device ensures that pharmaceutical products are never exposed to the environment during the testing process. This test system offers an optimized and fully regulatory compliant testing process when used with the Steritest® Symbio pump, specific accessories and high-quality culture media and rinsing fluids. The Steritest® NEO device for liquids in cartridge includes a short (20 mm) single needle for easy and safe access to cartridge or small soft plastic container and a separate vent needle. The red canister base indicates low adsorption Durapore® Poly vinylidene fluoride (PVDF) membrane and specific drain design. This optimizes the rinsing of products that inhibit the microbial growth.
Anwendung
Leistungsmerkmale und Vorteile
- One-stop-shop for sterility testing with our devices, pumps, media, fluids, and services
- Steritest® devices are manufactured in our Center of Excellence in Molsheim, France, with high-quality control standards maintaining the Certificate of Quality for each lot.
- New needle design
- Smarter workflow
- Completely closed set up
- Consistent performance
- New tubing disconnection tool
Verpackung
Rechtliche Hinweise
konfiguriert für
Analysenzertifikate (COA)
Suchen Sie nach Analysenzertifikate (COA), indem Sie die Lot-/Chargennummer des Produkts eingeben. Lot- und Chargennummern sind auf dem Produktetikett hinter den Wörtern ‘Lot’ oder ‘Batch’ (Lot oder Charge) zu finden.
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Verwandter Inhalt
Regulatory compliant membrane filtration sterility testing devices to ensure the safety of your pharmaceutical products.
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.