Wichtige Dokumente
718750
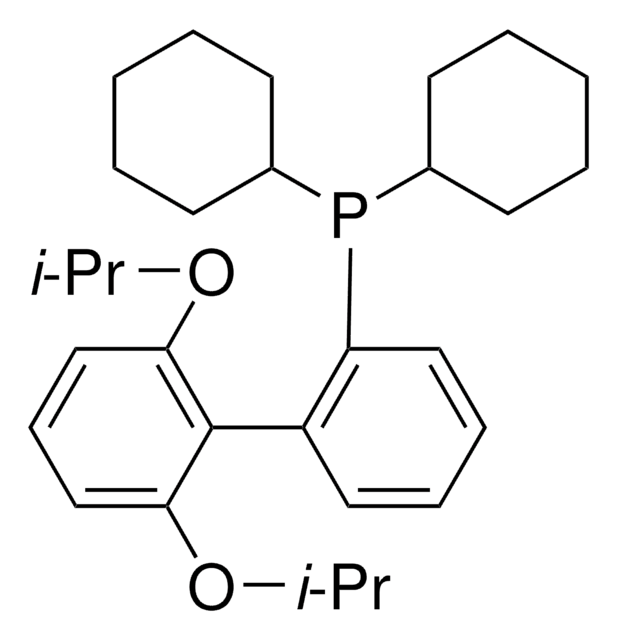
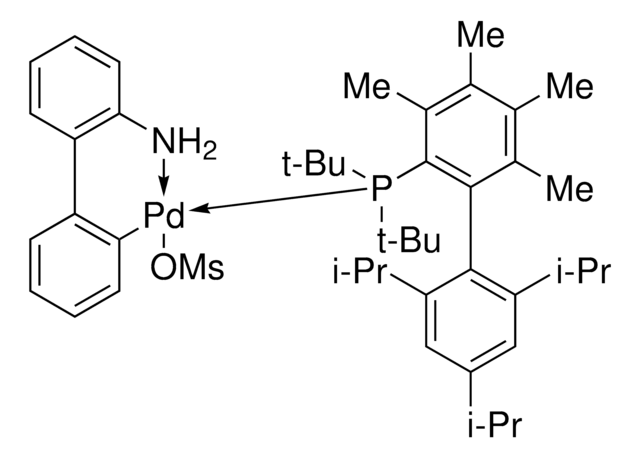
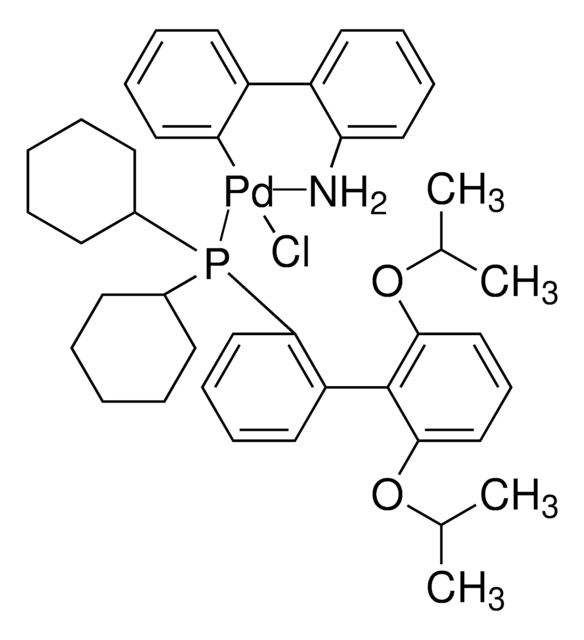
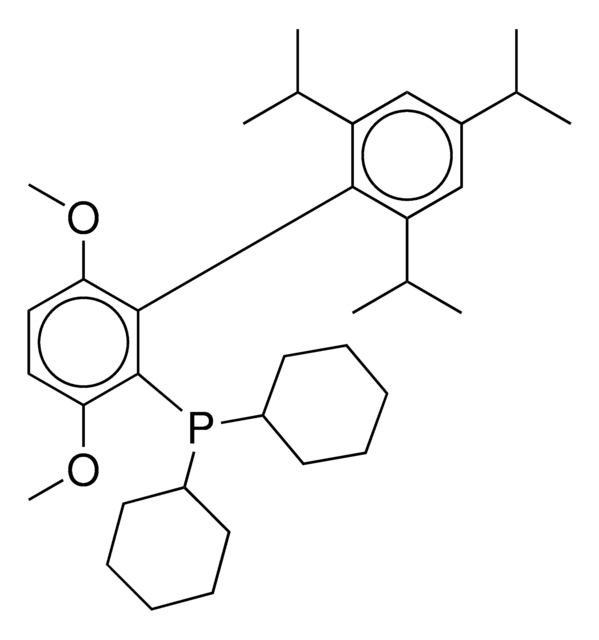
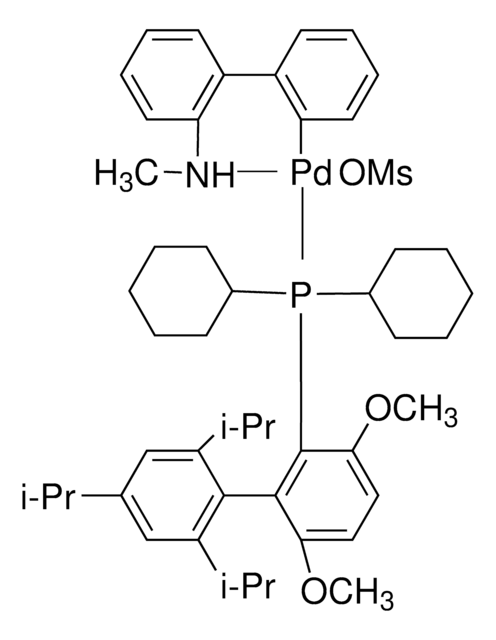
Chloro-[2-(dicyclohexylphosphino)-3,6-dimethoxy-2′,4′, 6′-triisopropyl-1,1′-biphenyl][2-(2-aminoethyl)-phenyl]-palladium(II)
may contain up to 1 mole equivalent of MTBE, 97%
Synonym(e):
(BrettPhos) palladium(II) phenethylamine chloride, BrettPhos Palladacycle, BrettPhos precatalyst, Chloro[2-(dicyclohexylphosphino)-3,6-dimethoxy-2′,4′, 6′-triisopropyl-1,1′-biphenyl][2-(2-aminoethyl)phenyl]palladium(II)
About This Item
Empfohlene Produkte
Qualitätsniveau
Assay
97%
Form
solid
Leistungsmerkmale
generation 1
Eignung der Reaktion
core: palladium
reaction type: Buchwald-Hartwig Cross Coupling Reaction
reaction type: Heck Reaction
reaction type: Hiyama Coupling
reaction type: Negishi Coupling
reaction type: Sonogashira Coupling
reaction type: Stille Coupling
reaction type: Suzuki-Miyaura Coupling
reagent type: catalyst
reaction type: Cross Couplings
mp (Schmelzpunkt)
198-204 °C
Funktionelle Gruppe
phosphine
SMILES String
COC(C)(C)C.NCCc1ccccc1[Pd]Cl.COc2ccc(OC)c(c2P(C3CCCCC3)C4CCCCC4)-c5c(cc(cc5C(C)C)C(C)C)C(C)C
InChI
1S/C35H53O2P.C8H10N.C5H12O.ClH.Pd/c1-23(2)26-21-29(24(3)4)33(30(22-26)25(5)6)34-31(36-7)19-20-32(37-8)35(34)38(27-15-11-9-12-16-27)28-17-13-10-14-18-28;9-7-6-8-4-2-1-3-5-8;1-5(2,3)6-4;;/h19-25,27-28H,9-18H2,1-8H3;1-4H,6-7,9H2;1-4H3;1H;/q;;;;+1/p-1
InChIKey
OWHWOTGYDWMPCA-UHFFFAOYSA-M
Allgemeine Beschreibung
Anwendung
Catalyst for:
- C,N-cross coupling of unprotected 3-halo-2-aminopyridines with primary and secondary amines
- Amination reaction
- N-arylation of aminophenols
Signalwort
Warning
H-Sätze
Gefahreneinstufungen
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Zielorgane
Respiratory system
Lagerklassenschlüssel
11 - Combustible Solids
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Kunden haben sich ebenfalls angesehen
Verwandter Inhalt
The Buchwald group has developed a series of highly active and versatile palladium precatalysts and biarylphosphine ligands used in cross-coupling reactions for the formation of C-C, C–N, C–O, C–F, C–CF3, and C–S bonds. The ligands are electron-rich, and highly tunable to provide catalyst systems with a diverse scope, high stability and reactivity. Furthermore, the new series of precatalysts are air-, moisture and thermally-stable and display good solubility in common organic solvents. The use of precatalysts ensures the efficient generation of the active catalytic species and allows one to accurately adjust the ligand:palladium ratio. The ligands, precatalysts and methodology developed in the Buchwald group are user friendly and have rendered previously difficult cross couplings reactions, much easier to achieve.
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.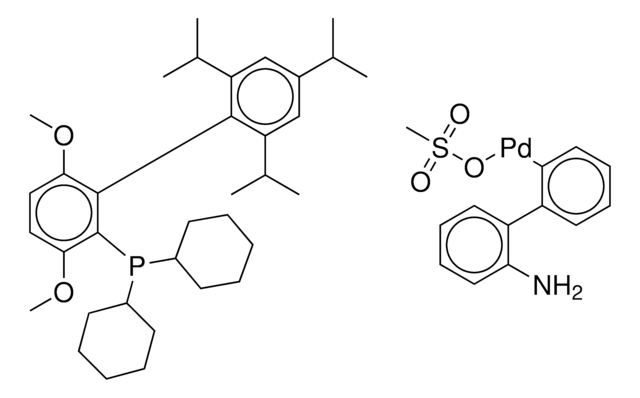

 95%](/deepweb/assets/sigmaaldrich/product/structures/151/609/eeb99dc1-9ef2-49d8-b255-6b5e2519fee1/640/eeb99dc1-9ef2-49d8-b255-6b5e2519fee1.png)