47636
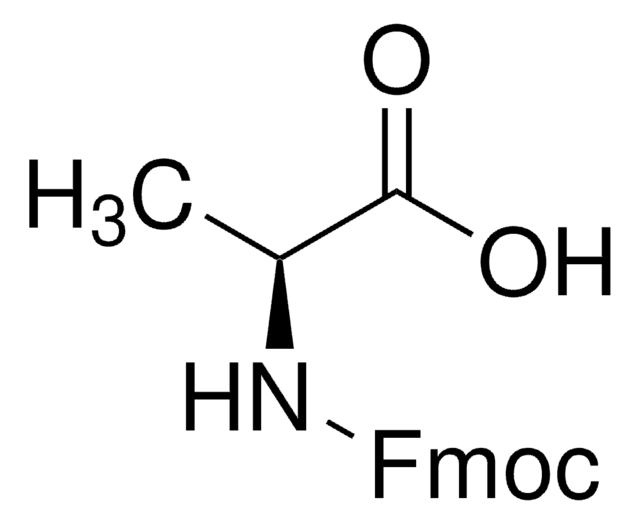
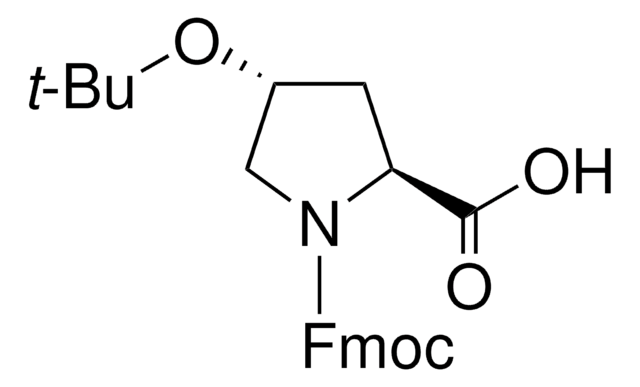
Fmoc-Pro-OH
≥90% (HPLC)
Synonym(e):
N-(Fluorenyl-9-methoxycarbonyl)-L-prolin, Fmoc-L-prolin
About This Item
Empfohlene Produkte
Qualitätsniveau
Assay
≥90% (HPLC)
Optische Aktivität
[α]20/D −32±1°, c = 1% in DMF
Eignung der Reaktion
reaction type: Fmoc solid-phase peptide synthesis
mp (Schmelzpunkt)
117-118 °C (lit.)
Anwendung(en)
peptide synthesis
Funktionelle Gruppe
Fmoc
Lagertemp.
2-8°C
SMILES String
OC(=O)[C@@H]1CCCN1C(=O)OCC2c3ccccc3-c4ccccc24
InChI
1S/C20H19NO4/c22-19(23)18-10-5-11-21(18)20(24)25-12-17-15-8-3-1-6-13(15)14-7-2-4-9-16(14)17/h1-4,6-9,17-18H,5,10-12H2,(H,22,23)/t18-/m0/s1
InChIKey
ZPGDWQNBZYOZTI-SFHVURJKSA-N
Suchen Sie nach ähnlichen Produkten? Aufrufen Leitfaden zum Produktvergleich
Lagerklassenschlüssel
11 - Combustible Solids
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Persönliche Schutzausrüstung
Eyeshields, Gloves, type N95 (US)
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
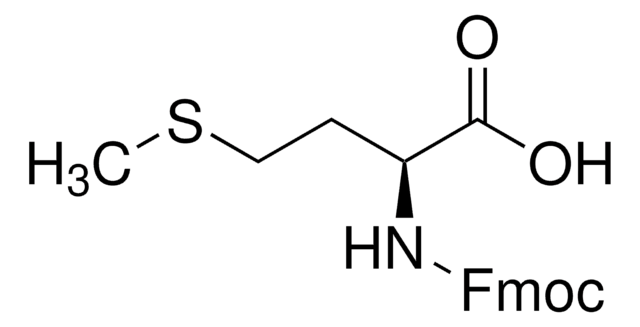
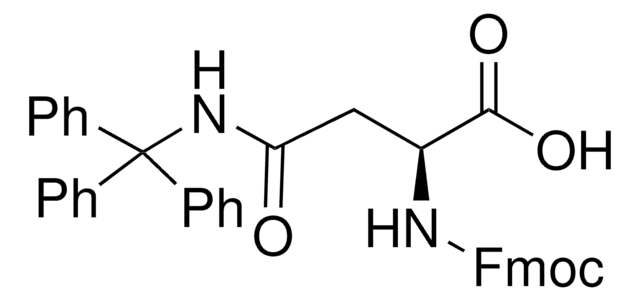
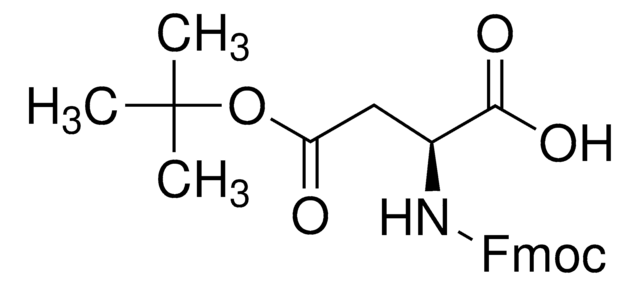
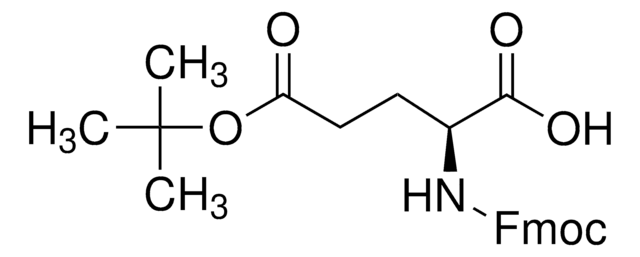
Kunden haben sich ebenfalls angesehen
Artikel
Proline analogues are promising candidates for tuning the biological, pharmaceutical, or physicochemical properties of naturally occuring, as well as de novo designed, linear, and, cyclic peptides.
With a growing peptide drug market the fast, reliable and uncomplicated synthesis of peptides is of paramount importance.
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.