190039
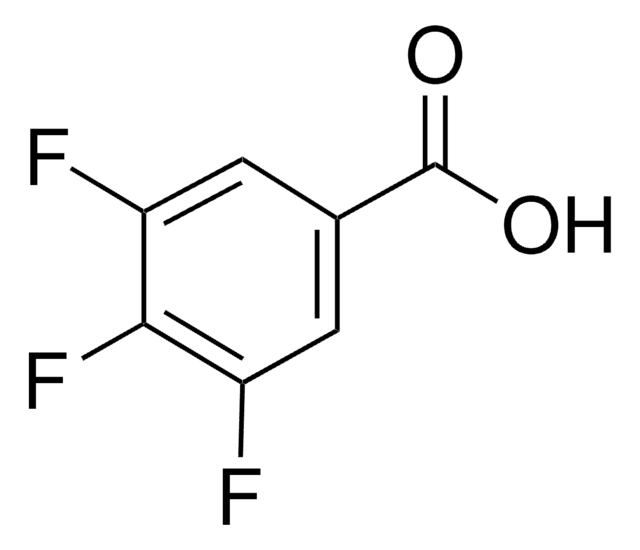
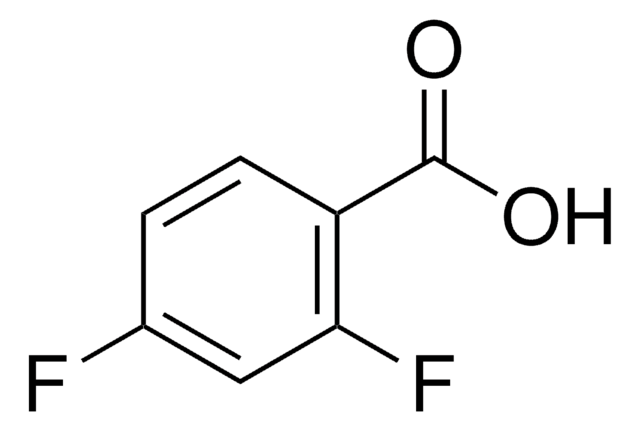
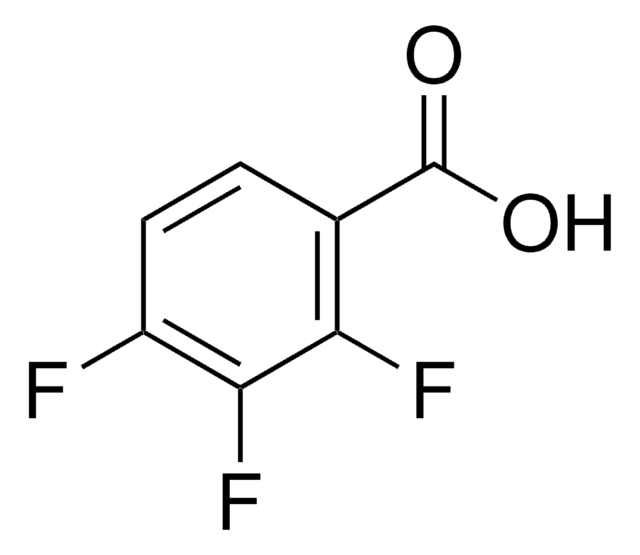
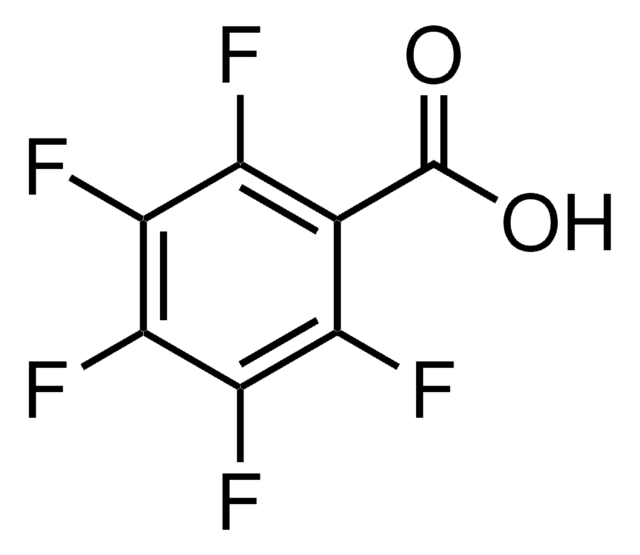
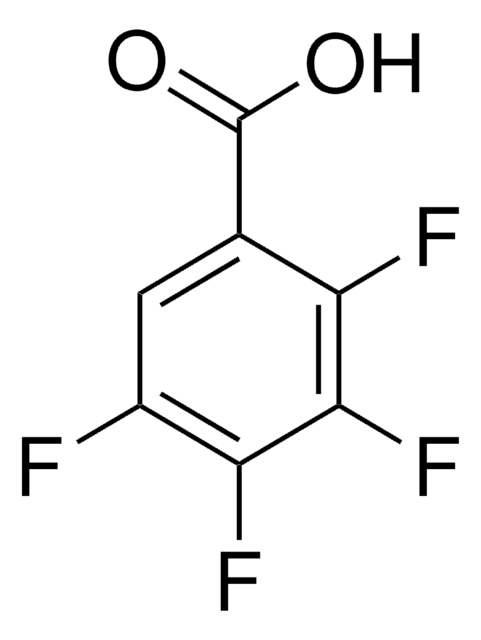
2,6-Difluorbenzoesäure
98%
Anmeldenzur Ansicht organisationsspezifischer und vertraglich vereinbarter Preise
Alle Fotos(2)
About This Item
Lineare Formel:
F2C6H3CO2H
CAS-Nummer:
Molekulargewicht:
158.10
Beilstein:
973774
EG-Nummer:
MDL-Nummer:
UNSPSC-Code:
12352100
PubChem Substanz-ID:
NACRES:
NA.22
Empfohlene Produkte
Qualitätsniveau
Assay
98%
Form
solid
mp (Schmelzpunkt)
157-161 °C (lit.)
Funktionelle Gruppe
carboxylic acid
fluoro
SMILES String
OC(=O)c1c(F)cccc1F
InChI
1S/C7H4F2O2/c8-4-2-1-3-5(9)6(4)7(10)11/h1-3H,(H,10,11)
InChIKey
ONOTYLMNTZNAQZ-UHFFFAOYSA-N
Suchen Sie nach ähnlichen Produkten? Aufrufen Leitfaden zum Produktvergleich
Verwandte Kategorien
Allgemeine Beschreibung
2,6-Difluorobenzoic acid is the major degradation product of diflubenzuron.
Anwendung
2,6-Difluorobenzoic acid has been used in the synthesis of 2,6-difluoro-N-(3-methoxy-1H-pyrazolo[3,4-b]pyridine-5-yl)-3-(propylsulfonamidio)benzamide and methyl 2,6-difluorobenzoate.
Signalwort
Warning
H-Sätze
Gefahreneinstufungen
Eye Irrit. 2 - Skin Irrit. 2
Lagerklassenschlüssel
11 - Combustible Solids
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Persönliche Schutzausrüstung
dust mask type N95 (US), Eyeshields, Gloves
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Kunden haben sich ebenfalls angesehen
E Gattavecchia et al.
Journal of environmental science and health. Part. B, Pesticides, food contaminants, and agricultural wastes, 16(2), 159-166 (1981-01-01)
Diflubenzuron (I) and its major degradation products 4-chlorophenyl urea (II), 2,6-difluorobenzoic acid (III) and 4-chloroaniline (IV) were tested for their activity on Euglena gracilis Z. The inhibition on the growth and on the incorporation of glycine-U-14C in the protein of
Li Chen et al.
Journal of agricultural and food chemistry, 53(1), 38-41 (2005-01-06)
Two series of benzoylphenylurea derivatives were synthesized as candidate propesticides by a nucleophilic addition reaction between 2,6-difluronbenzoyl isocyanate and N-substitutedaniline. The new compounds were identified by 1H NMR spectroscopy, electron ionization-mass spectrometry, and elemental analyses. The bioactivities of the new
A Karipides et al.
Acta crystallographica. Section C, Crystal structure communications, 48 ( Pt 6), 1015-1018 (1992-06-15)
Ca2+.2C7H3O2F2-.2H2O, M(r) = 390.3, monoclinic, C2/c, a = 17.584 (4), b = 10.771 (3), c = 7.887 (2) A, beta = 91.28 (2) degrees, V = 1493 A3, Z = 4, Dm = 1.75, Dx = 1.74 g cm-3, lambda(Mo
Min Wang et al.
Bioorganic & medicinal chemistry letters, 23(4), 1017-1021 (2013-01-09)
The authentic standard 2,6-difluoro-N-(3-methoxy-1H-pyrazolo[3,4-b]pyridine-5-yl)-3-(propylsulfonamidio)benzamide was synthesized from 2,6-difluorobenzoic acid and 3-amino-5-hydroxypyrazole in 9 steps with 1% overall chemical yield. Direct desmethylation of the reference standard with TMSCl/NaI gave the precursor 2,6-difluoro-N-(3-hydroxy-1H-pyrazolo[3,4-b]pyridine-5-yl)-3-(propylsulfonamidio)benzamide for radiolabeling in 70% yield. The target tracer 2,6-difluoro-N-(3-[(11)C]methoxy-1H-pyrazolo[3,4-b]pyridine-5-yl)-3-(propylsulfonamidio)benzamide
J Koerts et al.
Xenobiotica; the fate of foreign compounds in biological systems, 27(8), 801-817 (1997-08-01)
1. The metabolic fate of the insecticide teflubenzuron, orally dosed to the male Wistar rat, was investigated. Particular attention was paid to the metabolic fate of the benzoyl and aniline moiety after hydrolysis of the urea bridge. 2. The 0-48-h
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.