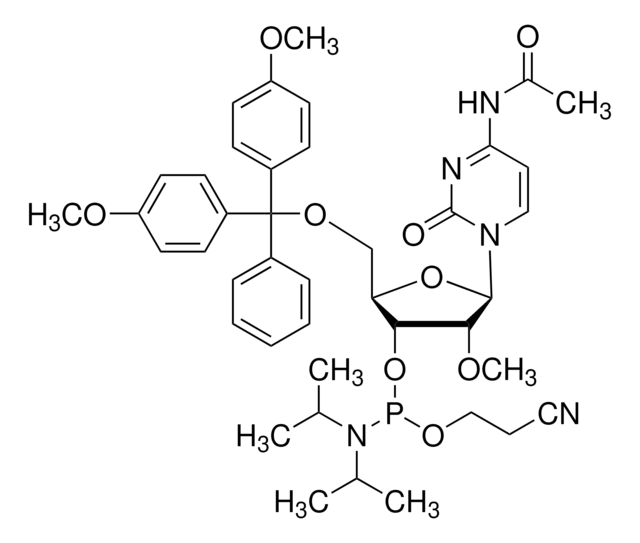
U211140
DMT-2′O-Methyl-rU Phosphoramidite
configured for MerMade
Sinonimo/i:
5′-O-[bis(4-methoxyphenyl)phenylmethyl]-2′-O-methyl-uridine, 3′-[2-cyanoethyl N,N-bis(1-methylethyl)phosphoramidite], DMT-2′O-Methyl-rU amidite
About This Item
Prodotti consigliati
Origine biologica
non-animal source (no BSE/TSE risk)
Livello qualitativo
Nome Commerciale
Proligo Reagents
Saggio
≥99% (31P-NMR)
≥99.0% (reversed phase HPLC)
Forma fisica
powder
PM
760.81 g/mol
Impurezze
≤0.3% mU2 (reversed phase HPLC, Hydrolysate)
≤0.3% mU3 (reversed phase HPLC, DMT-rme)
≤0.3% water content (Karl Fischer)
≤0.5% P(III) Impurities 100-169ppm (31P-NMR)
≤0.5% single Impurity (redox titration)
≤1.0% mU1 (reversed phase HPLC, DMT-rUme-DMT)
≤3% residual Solvent content
Colore
white to off-white
Compatibilità
conforms to structure for H-NMR
conforms to structure for LC-MS
configured for MerMade
Temperatura di conservazione
2-8°C
Stringa SMILE
CO[C@@H]1[C@H](OP(OCCC#N)N(C(C)C)C(C)C)[C@@H](COC(c2ccccc2)(c3ccc(OC)cc3)c4ccc(OC)cc4)O[C@H]1N5C=CC(=O)NC5=O
InChI
1S/C40H49N4O9P/c1-27(2)44(28(3)4)54(51-25-11-23-41)53-36-34(52-38(37(36)49-7)43-24-22-35(45)42-39(43)46)26-50-40(29-12-9-8-10-13-29,30-14-18-32(47-5)19-15-30)31-16-20-33(48-6)21-17-31/h8-10,12-22,24,27-28,34,36-38H,11,25-26H2,1-7H3,(H,42,45,46)/t34-,36-,37-,38-,54?/m1/s1
UVUOJOLPNDCIHL-XKZJCBTISA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Descrizione generale
- High yield of crude oligonucleotides Compatible with DNA synthesis
- Can be employed together with DNA or RNA phosphoramidites in the same synthesis to produce mixmer oligonucleotides
- Recommended deprotection conditions are 8 hours at 55 deg C using concentrated ammonia solution, or with AMA (concentrated ammonia/40% aqueous methylamine I/I, v/v) for 10 minutes at 65 deg C
- Purification and other downstream processing of fully modified 2′OMethyl RNA oligonucleotides are simpler than in the case of RNA, as no special precautions are required to provide protection against nucleolytic degradation
- Synthesis of 2′O-Methyl RNA oligonucleotides is similar to standard DNA synthesis but requires an elongated coupling time (recommended is 6minutes compared to 90 seconds for DNA monomers)
- 2′O-Methyl RNA phosphoramidites are also available with fast deprotection chemistry
Applicazioni
- Diagnostic probes
- Aptamer and ribozyme development
- Mixed 2′O-Methyl-RNA/DNA antisense molecules
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Certificati d'analisi (COA)
Cerca il Certificati d'analisi (COA) digitando il numero di lotto/batch corrispondente. I numeri di lotto o di batch sono stampati sull'etichetta dei prodotti dopo la parola ‘Lotto’ o ‘Batch’.
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.