T1783
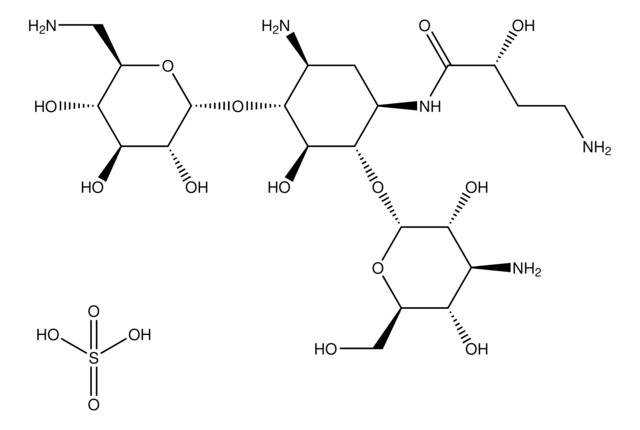
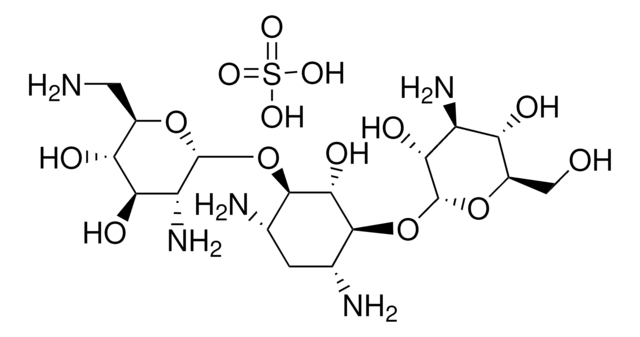
Tobramycin sulfate salt
aminoglycoside antibiotic
Sinonimo/i:
Nebramycin Factor 6 sulfate
About This Item
Prodotti consigliati
Origine biologica
Streptomyces tenebrarius
Livello qualitativo
Stato
powder
Potenza
634-739 μg per mg
Condizioni di stoccaggio
(Keep container tightly closed in a dry and well-ventilated place. Hygroscopic.)
Colore
white to off-white
Solubilità
H2O: soluble 50 mg/mL
Spettro attività antibiotica
Gram-negative bacteria
Modalità d’azione
protein synthesis | interferes
Temperatura di conservazione
2-8°C
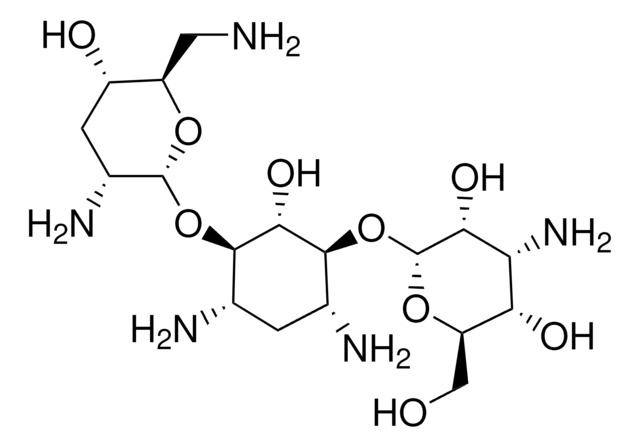
Stringa SMILE
OS(O)(=O)=O.NC[C@H]1O[C@H](O[C@@H]2[C@@H](N)C[C@@H](N)[C@H](O[C@H]3O[C@H](CO)[C@@H](O)[C@H](N)[C@H]3O)[C@H]2O)[C@H](N)C[C@@H]1O
InChI
1S/C18H37N5O9.H2O4S/c19-3-9-8(25)2-7(22)17(29-9)31-15-5(20)1-6(21)16(14(15)28)32-18-13(27)11(23)12(26)10(4-24)30-18;1-5(2,3)4/h5-18,24-28H,1-4,19-23H2;(H2,1,2,3,4)/t5-,6+,7+,8-,9+,10+,11-,12+,13+,14-,15+,16-,17+,18+;/m0./s1
ZEUUPKVZFKBXPW-TWDWGCDDSA-N
Categorie correlate
Descrizione generale
Applicazioni
Azioni biochim/fisiol
Mode of Action: Binds to 70S ribosomal subunit; inhibits translocation; elicits miscoding.
Spectrum of Activity: Gram negative bacteria. Not effective against Enterococci.
Confezionamento
Avvertenza
Altre note
Avvertenze
Danger
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Acute Tox. 4 Dermal - Acute Tox. 4 Inhalation - Repr. 1B
Codice della classe di stoccaggio
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
Eyeshields, Gloves, type P3 (EN 143) respirator cartridges
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Articoli
Extraction and quantitative analysis of aminoglycosides in porcine tissue, using molecular imprinted polymer solid phase extraction followed by LC-MS/MS.
Protein synthesis is a complex, multi-step process involving many enzymes as well as conformational alignment. However, the majority of antibiotics that block bacterial protein synthesis interfere with the processes at the 30S subunit or 50S subunit of the 70S bacterial ribosome.
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.