O4126
Oxaloacetic acid
≥97% (HPLC)
Sinonimo/i:
2-Oxosuccinic acid, Ketosuccinic acid, Oxalacetic acid, Oxobutanedioic acid
About This Item
Prodotti consigliati
Livello qualitativo
Saggio
≥97% (HPLC)
Stato
powder
Solubilità
H2O: 100 mg/mL, clear to slightly hazy, colorless to light yellow
Temperatura di conservazione
−20°C
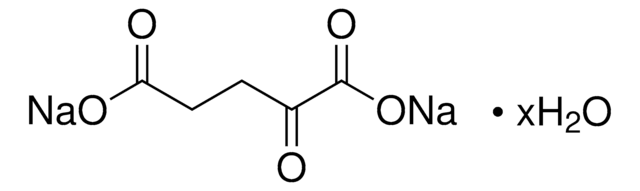
Stringa SMILE
OC(=O)CC(=O)C(O)=O
InChI
1S/C4H4O5/c5-2(4(8)9)1-3(6)7/h1H2,(H,6,7)(H,8,9)
KHPXUQMNIQBQEV-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Descrizione generale
Applicazioni
Azioni biochim/fisiol
Avvertenze
Warning
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Eye Irrit. 2
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 1
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Articoli
Learn about monosaccharide biosynthesis and the metabolism of monosaccharides. A unit of a carbohydrate and the simplest form of a sugar, a monosaccharide cannot be hydrolyzed into a simpler compound.
Sigma-Aldrich presents an article about how proliferatively active cells require both a source of carbon and of nitrogen for the synthesis of macromolecules. Although a large proportion of tumor cells utilize aerobic glycolysis and shunt metabolites away from mitochondrial oxidative phosphorylation, many tumor cells exhibit increased mitochondrial activity.
Information on fatty acid synthesis and metabolism in cancer cells. Learn how proliferatively active cells require fatty acids for functions such as membrane generation, protein modification, and bioenergetic requirements. These fatty acids are derived either from dietary sources or are synthesized by the cell.
Protocolli
Enzymatic Assay of Malic Dehydrogenase (EC 1.1.1.37)
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.