G0413
β(1→4)-Galactosidase, positionally specific from Streptococcus pneumoniae
recombinant, expressed in E. coli, buffered aqueous solution
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Numero CAS:
Numero MDL:
Codice UNSPSC:
12352204
NACRES:
NA.32
Prodotti consigliati
Ricombinante
expressed in E. coli
Livello qualitativo
Forma fisica
buffered aqueous solution
Attività specifica
≥6 units/mg protein
Confezionamento
vial of 0.06 unit
N° accesso UniProt
Condizioni di spedizione
wet ice
Temperatura di conservazione
2-8°C
Informazioni sul gene
human ... GLB1(2720)
Categorie correlate
Descrizione generale
β-Galactosidase is present in bacteria, fungi, yeast and animal organs. It is also found in fruits, such as apples, almonds and apricots. β-Galactosidase is a tetramer and is made up of four polypeptide chains consisting of amino acids that assemble to form five structural domains. The domains are jelly roll barrel, a central domain that serves as an active site and the remaining domains are composed of β-sandwich and fibronectin.
Applicazioni
β(1→4)-Galactosidase, positionally specific from Streptococcus pneumonia has been used:
- as a position-specific enzyme to study its effects in the terminal galactosylation with protective efficacy of glycosphingolipid (GSPL) in mice.
- for the digestion of radioactive oligosaccharides.
- as a position-specific enzymeto study its effects on the virulence profile of avirulent Leishmania donovani clone (A-LD).
Azioni biochim/fisiol
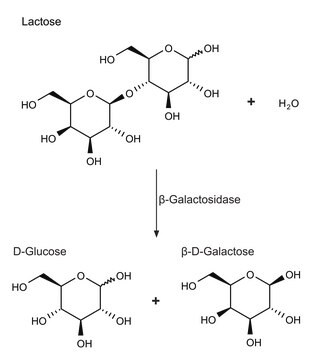
β-Galactosidase plays a role in hydrolyzing the D-galactosyl moieties in oligosaccharides, polymers and secondary metabolites. It is widely applicable in the dairy industry to remove lactose from milk and dairy products for the benefit of lactose-intolerant individuals. β-Galactosidase is also applicable in the food industry to improve the sweetness, flavor and solubility.
Definizione di unità
One unit will hydrolyze 1 μmole of p-nitrophenyl β-D-galactopyranoside per min at pH 5.0 at 37 °C.
Stato fisico
Solution in 20 mM Tris-HCl, pH 7.5, 25 mM NaCl
Codice della classe di stoccaggio
10 - Combustible liquids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Certificati d'analisi (COA)
Cerca il Certificati d'analisi (COA) digitando il numero di lotto/batch corrispondente. I numeri di lotto o di batch sono stampati sull'etichetta dei prodotti dopo la parola ‘Lotto’ o ‘Batch’.
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
Virulence attenuation of a UDP-galactose/N-acetylglucosamine beta1, 4 galactosyltransferase expressing Leishmania donovani promastigote
Bhaumik, SK , et al.
Glycoconjugate Journal, 25(5), 459-472 (2008)
Shaima Saqib et al.
3 Biotech, 7(1), 79-79 (2017-05-14)
The enzyme β-galactosidases have been isolated from various sources such as bacteria, fungi, yeast, vegetables, and recombinant sources. This enzyme holds importance due to its wide applications in food industries to manufacture lactose-hydrolyzed products for lactose-intolerant people and the formation
TLR4 and NKT cell synergy in immunotherapy against visceral leishmaniasis
Karmakar S, et al.
PLoS Pathogens, 8(4), 79-79 (2012)
S K Bhaumik et al.
Glycoconjugate journal, 25(5), 459-472 (2008-01-17)
Protozoan parasites of the genus Leishmania are the causative agent of leishmaniasis, a disease whose manifestations in humans range from mild cutaneous lesions to fatal visceral infections. Human visceral leishmaniasis is caused by Leishmania donovani. Long-term culture in vitro leads
beta Galactosidases and their potential applications: a review
Husain Q
Critical Reviews in Biotechnology, 30(1), 41-62 (2010)
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.