HLINJMAG-75K
MILLIPLEX® Human Liver Injury Magnetic Bead Panel - Toxicity Multiplex Assay
About This Item
Prodotti consigliati
Descrizione
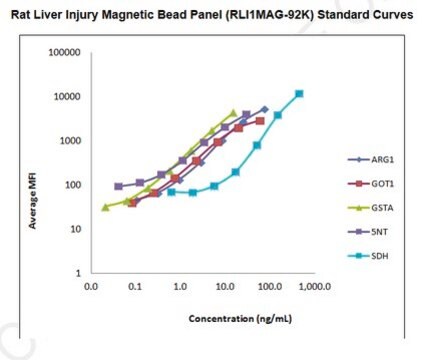
The analytes available for this multiplex kit are: Liver-type arginase 1 (ARG1), α-glutathianone S-transferase (GSTα), malate dehydrogenase 1 (MDH1), sorbitol dehydrogenase (SDH), and 5′-Nucleotidase (5′-NT/CD73).
Livello qualitativo
100
200
Reattività contro le specie
human
Produttore/marchio commerciale
Milliplex®
assay range
accuracy: 106-123%
standard curve range: 14-10,000 pg/mL
(GSTα)
standard curve range: 41-30,000 pg/mL
(5′-NT/CD73)
standard curve range: 549-400,000 pg/mL
(SDH)
standard curve range: 69-50,000 pg/mL
(ARG1)
standard curve range: 69-50,000 pg/mL
(MDH1)
tecniche
multiplexing: suitable
Metodo di rivelazione
fluorometric (Luminex xMAP)
Condizioni di spedizione
ambient
Categorie correlate
Descrizione generale
The MILLIPLEX® Human Liver Injury Panel contains all the components necessary to simultaneous quantify the following 5 analytes in serum and plasma samples:
• Liver-Type Arginase 1 (ARG1)*
• Malate dehydrogenase 1 (MDH1)*
• α-glutathione S-transferase (GSTα)*
• Sorbitol Dehydrogenase (SDH)*
• 5′-Nucleotidase/CD73 (5′-NT)
*ARG1, GSTα, and SDH are biomarkers listed in the Predictive Safety Testing Consortium (PSTC) project pipeline which have a strong translational role in drug safety testing.
The MILLIPLEX® portfolio offers the broadest selection of analytes across a wide range of disease states and species. Once the analytes of interest have been identified, you can rely on the quality that we build into each kit to produce results you can trust. In addition to the assay characteristics listed in the protocol, other performance criteria evaluated during the validation process include: cross-reactivity, dilution linearity, kit stability, and sample behavior (e.g. detectability and stability).
Panel Type: Toxicity
Specificità
There was no or negligible cross-reactivity between the antibodies for an analyte and any of the other analytes within a panel.
Applicazioni
- Analytes: ARG1, GSTα, SDH 5′-NT/CD73, Malate dehydrogenase 1 (MDH1)
- Recommended Sample type: serum, plasma, and tissue culture supernatants
- Recommended Sample dilution: 1:5 serum or plasma samples. Tissue culture supernatants may require dilution as well.
- Assay Run Time: One day or Overnight
- Research Category: Toxicity
Caratteristiche e vantaggi
Confezionamento
Stoccaggio e stabilità
Altre note
Note legali
Esclusione di responsabilità
Certificati d'analisi (COA)
Cerca il Certificati d'analisi (COA) digitando il numero di lotto/batch corrispondente. I numeri di lotto o di batch sono stampati sull'etichetta dei prodotti dopo la parola ‘Lotto’ o ‘Batch’.
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.