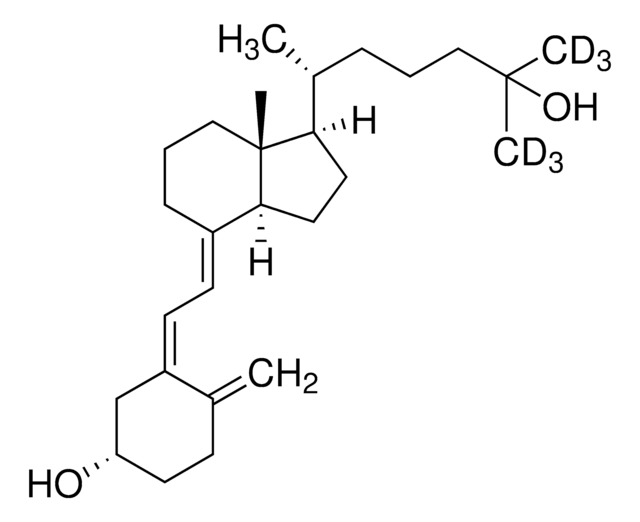
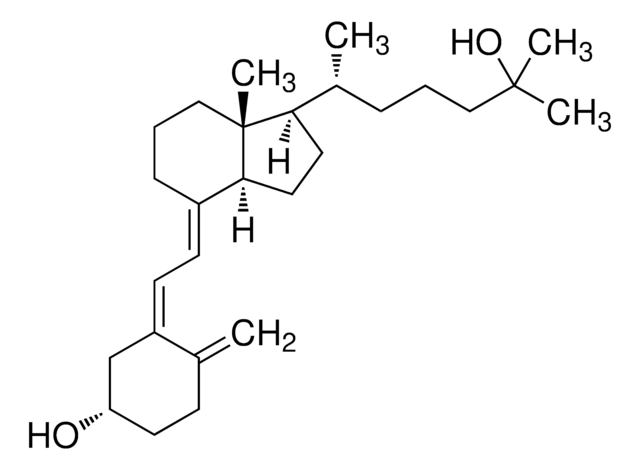
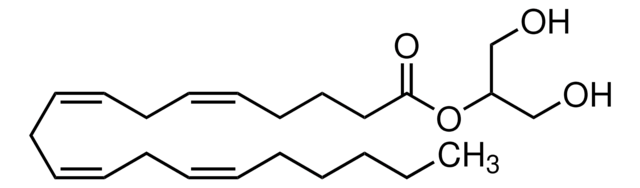
870450O
Avanti
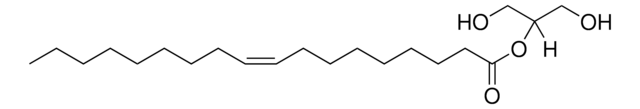
2-AG
Avanti Research™ - A Croda Brand 870450O
Sinonimo/i:
2-arachidonoyl glycerol
About This Item
Prodotti consigliati
Forma fisica
liquid
Confezionamento
pkg of 1 × 5 mg (with screw cap/argon/foil bag (870450O-5mg))
Produttore/marchio commerciale
Avanti Research™ - A Croda Brand 870450O
Tipo di lipide
bioactive lipids
phosphoglycerides
Condizioni di spedizione
dry ice
Temperatura di conservazione
−70°C
Stringa SMILE
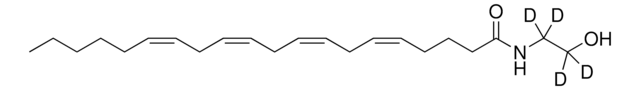
[H]C(CO)(OC(CCC/C=C\C/C=C\C/C=C\C/C=C\CCCCC)=O)CO
InChI
1S/C23H38O4/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-16-17-18-19-23(26)27-22(20-24)21-25/h6-7,9-10,12-13,15-16,22,24-25H,2-5,8,11,14,17-21H2,1H3/b7-6-,10-9-,13-12-,16-15-
RCRCTBLIHCHWDZ-DOFZRALJSA-N
Categorie correlate
Descrizione generale
Confezionamento
Note legali
Codice della classe di stoccaggio
12 - Non Combustible Liquids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Certificati d'analisi (COA)
Cerca il Certificati d'analisi (COA) digitando il numero di lotto/batch corrispondente. I numeri di lotto o di batch sono stampati sull'etichetta dei prodotti dopo la parola ‘Lotto’ o ‘Batch’.
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.