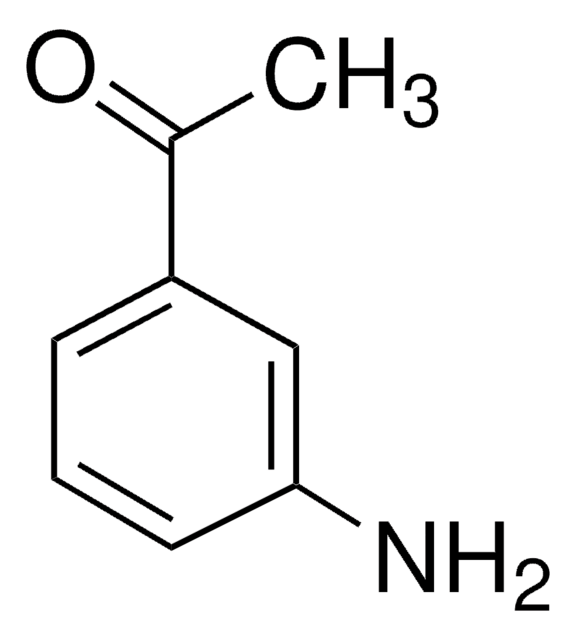
A38002
4′-Aminoacetophenone
99%
Sinonimo/i:
4-Acetylaniline
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula condensata:
H2NC6H4COCH3
Numero CAS:
Peso molecolare:
135.16
Beilstein:
471493
Numero CE:
Numero MDL:
Codice UNSPSC:
12352100
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Saggio
99%
Stato
powder
P. ebollizione
293 °C (lit.)
Punto di fusione
103-107 °C (lit.)
Stringa SMILE
CC(=O)c1ccc(N)cc1
InChI
1S/C8H9NO/c1-6(10)7-2-4-8(9)5-3-7/h2-5H,9H2,1H3
GPRYKVSEZCQIHD-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Categorie correlate
Avvertenze
Warning
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Acute Tox. 4 Oral
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
dust mask type N95 (US), Eyeshields, Faceshields, Gloves
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Anne Ly et al.
Organic & biomolecular chemistry, 3(5), 917-923 (2005-02-26)
The most easily oxidized sites in DNA are the guanine bases, and major intermediates produced by the direct effect of ionizing radiation (ionization of the DNA itself) are electron deficient guanine species. By means of a radiation chemical method (gamma-irradiation
Dominik Cincić et al.
Acta crystallographica. Section C, Crystal structure communications, 64(Pt 4), o226-o229 (2008-04-09)
In the title compounds, 4-carboxyanilinium bromide, C(7)H(8)NO(2)(+) x Br(-), (I), and 4-acetylanilinium bromide, C(8)H(10)NO(+) x Br(-), (II), each asymmetric unit contains a discrete cation with a protonated amino group and a halide anion. Both crystal structures are characterized by two-dimensional
O A Aleksintseva et al.
Antibiotiki, 27(7), 493-495 (1982-01-01)
The effect of p-aminobenzoic acid on the biosynthesis of levorin was studied. It was shown that in the presence of exogenic p-aminobenzoic acid the antibiotic activity increased by 11 per cent. The acid added was transformed into p-aminoacetophenone which was
O A Aleksintseva et al.
Antibiotiki, 26(8), 566-570 (1981-08-01)
A method for spectrophotometric determination of p-aminoacetophenone (p-AAP) in the mycelium and fermentation broth filtrates of organisms producing polyenic macrolide antibiotics is described. The level of p-AAP accumulation was studied as applicable to the biosynthesis of levorin, a polyenic antibiotic
O Raatikainen et al.
Journal of chromatography, 585(2), 247-254 (1991-11-01)
A high-performance liquid chromatographic (HPLC) method for the determination of the aromaticity of heptaene polyene antibiotics has been developed. The released aromatic moiety of the heptaene polyenes aureofungin, candicidin, candimycin, hamycin and trichomycin was assayed after alkaline hydrolysis. The presence
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.