803340
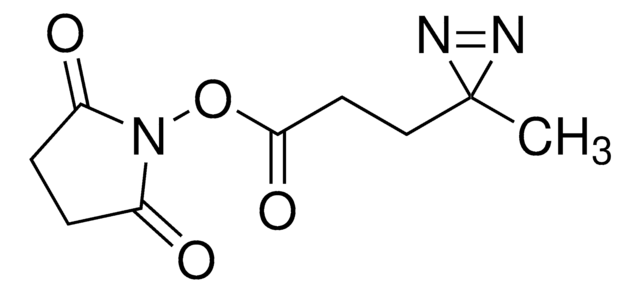
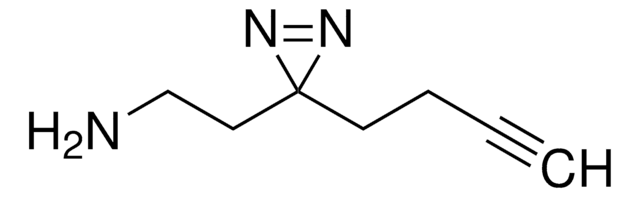
Sulfo-SDA (Sulfo-NHS-Diazirine) (sulfosuccinimidyl 4,4′-azipentanoate)
About This Item
Prodotti consigliati
Saggio
≥90%
Livello qualitativo
Stato
powder
PM
327.25
Impiego in reazioni chimiche
reagent type: cross-linking reagent
Condizioni di stoccaggio
desiccated
Solubilità
water: soluble
Condizioni di spedizione
ambient
Temperatura di conservazione
2-8°C
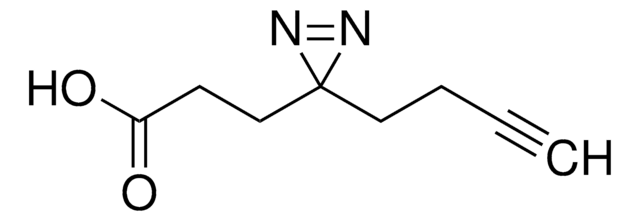
Stringa SMILE
O=C(C(S(=O)([O-])=O)C1)N(OC(CCC2(N=N2)C)=O)C1=O.[Na+]
InChI
1S/C9H11N3O7S.Na/c1-9(10-11-9)3-2-7(14)19-12-6(13)4-5(8(12)15)20(16,17)18;/h5H,2-4H2,1H3,(H,16,17,18);/q;+1/p-1
KTYCFZFVXSHAGH-UHFFFAOYSA-M
Categorie correlate
Descrizione generale
Caratteristiche e vantaggi
- Water soluble—solubility in aqueous solutions improved by a sulfonate group
- Heterobifunctional—NHS ester group reacts with primary amines at pH 7 to 9 to form covalent amide bonds; diazirine (azipentanoate) group reacts efficiently with any amino acid side chain or peptide backbone upon activation with long-wave UV light (330-370 nm)
- Controllable—two-step chemical crosslinking is activated using common laboratory UV lamps
- Easy to use—these crosslinkers are photo-stable under typical laboratory lighting conditions so there is no need to perform experiments in the dark
- Better than aryl azides—the diazirine photoreactive group has better photostability in normal light than phenyl azide groups of traditional photoreactive crosslinkers, yet the diazirine group is more efficiently activated by long-wave UV light
Avvertenza
Prodotti correlati
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Scegli una delle versioni più recenti:
Certificati d'analisi (COA)
It looks like we've run into a problem, but you can still download Certificates of Analysis from our Documenti section.
Se ti serve aiuto, non esitare a contattarci Servizio Clienti
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 803340-50MG | 4061832874630 |
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.