523429
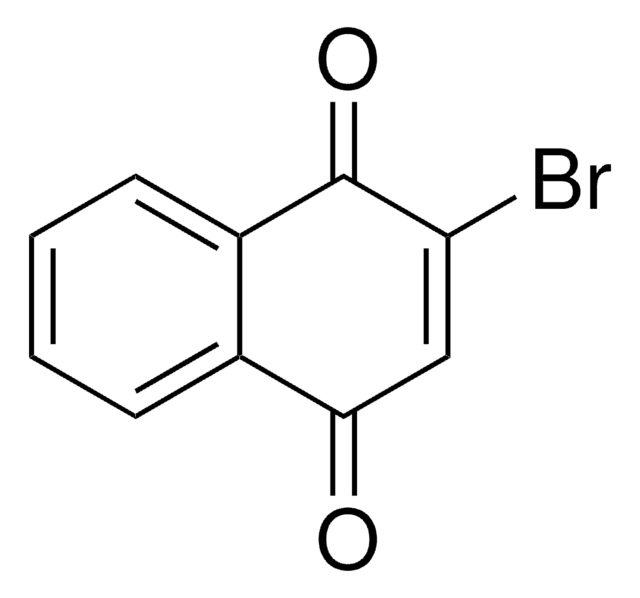
2,3-Dibromo-1,4-naphthoquinone
97%
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula empirica (notazione di Hill):
C10H4Br2O2
Numero CAS:
Peso molecolare:
315.95
Numero CE:
Numero MDL:
Codice UNSPSC:
12352100
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Livello qualitativo
Saggio
97%
Punto di fusione
218-222 °C (lit.)
Gruppo funzionale
bromo
ketone
Stringa SMILE
BrC1=C(Br)C(=O)c2ccccc2C1=O
InChI
1S/C10H4Br2O2/c11-7-8(12)10(14)6-4-2-1-3-5(6)9(7)13/h1-4H
PSMABVOYZJWFBV-UHFFFAOYSA-N
Categorie correlate
Descrizione generale
2,3-Dibromo-1,4-naphthoquinone is a 2,3-disubstituted 1,4-naphthoquinone. It is a plumbagin derivative and an acaricide. It undergoes photochemical reaction with 2-methoxy-1-alkene to yield derivatives of 2-(2-alkanonyl)-1,4-naphthoquinone.
Applicazioni
2,3-Dibromo-1,4-naphthoquinone may be used in the synthesis of 3-[3-(2-carboxy-ethylsulfanyl)-1,4-dioxo-1,4-dihydro-naphthalen-2-ylsulfanyl]-propionic acid and NSC 95397 (a protein tyrosine phosphatase antagonist).
Avvertenze
Warning
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Organi bersaglio
Respiratory system
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
dust mask type N95 (US), Eyeshields, Gloves
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
A new method of ?-keto alkyl chain introduction into 1, 4-naphthoquinone.
Maruyama K, et al.
Chemistry Letters (Jpn), 13(3), 371-374 (1984)
Chi-Hoon Lee et al.
Journal of microbiology and biotechnology, 18(2), 314-321 (2008-03-01)
Acaricidal effects of materials derived from Diospyros kaki roots against Dermatophagoides farinae and D. pteronyssinus were assessed using impregnated fabric disk bioassay and compared with that of the commercial benzyl benzoate. The observed responses varied according to dosage and mite
A one-pot synthesis of 1, 4-naphthoquinone-2, 3-bis-sulfides catalysed by a commercial laccase.
Wellington KW, et al.
Green Chemistry, 14(9), 2567-2576 (2012)
Vincent P Peyregne et al.
Molecular cancer therapeutics, 4(4), 595-602 (2005-04-14)
Cdc25 phosphatases are important in cell cycle control and activate cyclin-dependent kinases (Cdk). Efforts are currently under way to synthesize specific small-molecule Cdc25 inhibitors that might have anticancer properties. NSC 95397, a protein tyrosine phosphatase antagonist from the National Cancer
Xiao-Fei Shang et al.
Scientific reports, 8(1), 1609-1609 (2018-01-27)
As important secondary plant metabolites, naphthoquinones exhibit a wide range of biological activities. However, their potential as sustainable alternatives to synthetic acaricides has not been studied. This study for the first time investigates the acaricidal activity of naphthoquinones against Psoroptes
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.