460990
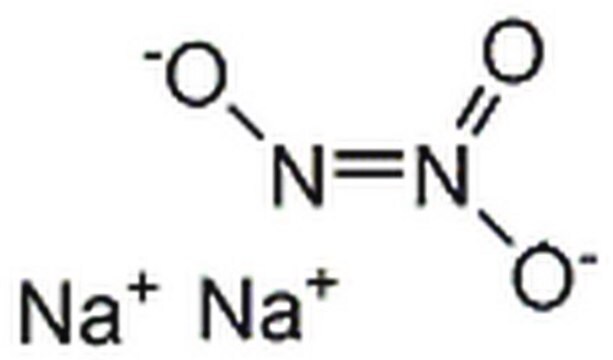
Sodium trans-hyponitrite hydrate
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula condensata:
Na2N2O2 · xH2O
Numero CAS:
Peso molecolare:
105.99 (anhydrous basis)
Numero MDL:
Codice UNSPSC:
12352302
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Applicazioni
Precursor to tert-butyl hyponitrite which was employed in the low-temperature thermal generation of tert-butoxy radicals.
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
Jun Wang et al.
Journal of the American Chemical Society, 131(2), 450-451 (2008-12-23)
A iron-dinitrosyl species ((6)L)Fe(NO)(2) (2), generated from nitrogen monoxide (*NO) binding to its related iron(II)-mononitrosyl complex ((6)L)Fe(NO) (1), efficiently effects reductive coupling of two *NO molecules to release nitrous oxide (N(2)O), when Cu(+) ion and 2 equiv acid are added;
The structure of the hyponitrite species in a heme Fe-Cu binuclear center.
Constantinos Varotsis et al.
Angewandte Chemie (International ed. in English), 46(13), 2210-2214 (2007-02-14)
Gregory A Poskrebyshev et al.
Journal of the American Chemical Society, 126(3), 891-899 (2004-01-22)
All major properties of the aqueous hyponitrite radicals (ONNO- and ONNOH), the adducts of nitric oxide (NO) and nitroxyl (3NO- and 1HNO), are revised. In this work, the radicals are produced by oxidation of various hyponitrite species in the 2-14
Nan Xu et al.
Inorganic chemistry, 49(14), 6253-6266 (2010-07-30)
Nitric oxide (NO) and its derivatives such as nitrite and hyponitrite are biologically important species of relevance to human health. Much of their physiological relevance stems from their interactions with the iron centers in heme proteins. The chemical reactivities displayed
Nan Xu et al.
Journal of the American Chemical Society, 131(7), 2460-2461 (2009-02-05)
The coupling of two nitric oxide (NO) molecules in heme active sites is an important contributor to the conversion of NO to nitrous oxide (N(2)O) by heme-containing enzymes. Several formulations for the presumed heme-Fe{N(2)O(2)}(n-) intermediates have been proposed previously, however
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.