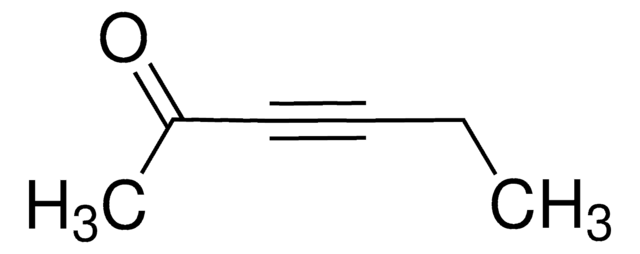418676
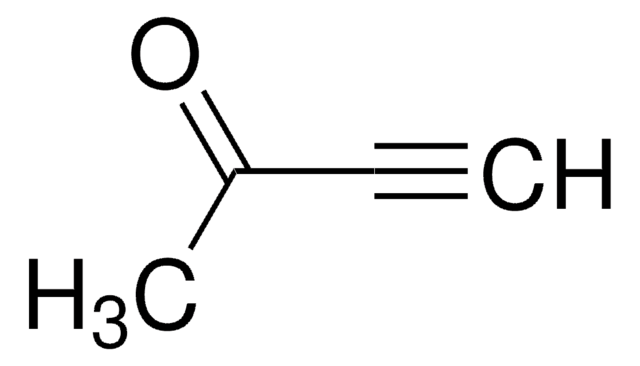
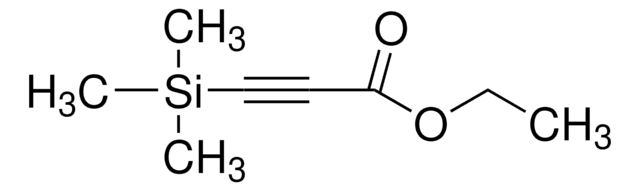
4-(Trimethylsilyl)-3-butyn-2-one
97%
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
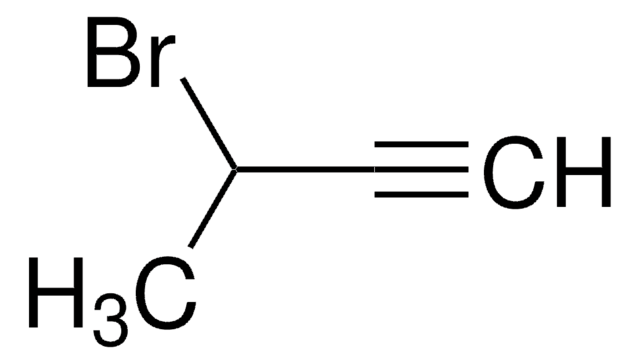
Formula condensata:
(CH3)3SiC≡CCOCH3
Numero CAS:
Peso molecolare:
140.26
Numero MDL:
Codice UNSPSC:
12352100
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Livello qualitativo
Saggio
97%
Stato
liquid
Indice di rifrazione
n20/D 1.442 (lit.)
P. ebollizione
156 °C (lit.)
Densità
0.854 g/mL at 25 °C (lit.)
Gruppo funzionale
ketone
Stringa SMILE
CC(=O)C#C[Si](C)(C)C
InChI
1S/C7H12OSi/c1-7(8)5-6-9(2,3)4/h1-4H3
NQEZDDPEJMKMOS-UHFFFAOYSA-N
Categorie correlate
Descrizione generale
4-(Trimethylsilyl)-3-butyn-2-one is a ketone. Its asymmetric bioreduction to enantiopure {(S)-TMSBOL in various hydrophilic ionic liquid (ILs) solvent systems has been reported.
Applicazioni
4-(Trimethylsilyl)-3-butyn-2-one (TMSB) has been used to investigate its asymmetric bioreduction to (S)-4-(trimethylsilyl)-3-butyn-2-ol {(S)-TMSBOL} by employing biocompatible water-immiscible ionic liquids (ILs). TMSB may be used for the synthesis of entecavir (BMS-200475).
Avvertenze
Warning
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Eye Irrit. 2 - Flam. Liq. 3 - Skin Irrit. 2 - STOT SE 3
Organi bersaglio
Respiratory system
Codice della classe di stoccaggio
3 - Flammable liquids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
82.4 °F - closed cup
Punto d’infiammabilità (°C)
28 °C - closed cup
Dispositivi di protezione individuale
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
Javier Velasco et al.
The Journal of organic chemistry, 78(11), 5482-5491 (2013-05-18)
Entecavir (BMS-200475) was synthesized from 4-trimethylsilyl-3-butyn-2-one and acrolein. The key features of its preparation are: (i) a stereoselective boron-aldol reaction to afford the acyclic carbon skeleton of the methylenecylopentane moiety; (ii) its cyclization by a Cp2TiCl-catalyzed intramolecular radical addition of
Wen-Yong Lou et al.
BMC biotechnology, 9, 90-90 (2009-10-24)
Whole cells are usually employed for biocatalytic reduction reactions to ensure efficient coenzyme regeneration and to avoid problems with enzyme purification and stability. The efficiency of whole cell-catalyzed bioreduction is frequently restricted by pronounced toxicity of substrate and/or product to
Bo-Bo Zhang et al.
PloS one, 7(5), e37641-e37641 (2012-06-05)
Hydrophilic ionic liquids (ILs) were employed as green solvents to construct an IL-containing co-solvent system for improving the asymmetric reduction of 4-(trimethylsilyl)-3-butyn-2-one by immobilized Candida parapsilosis cells. Among 14 hydrophilic ILs examined, 1-(2'-hydroxyl)ethyl-3-methylimidazolium nitrate (C(2)OHMIM·NO(3)) was considered as the most
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 418676-5G | 4061837581533 |
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.