370215
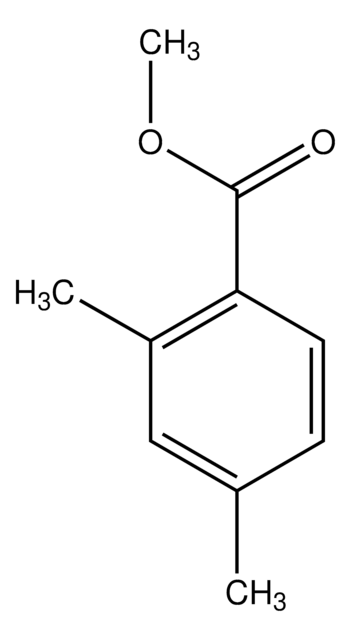
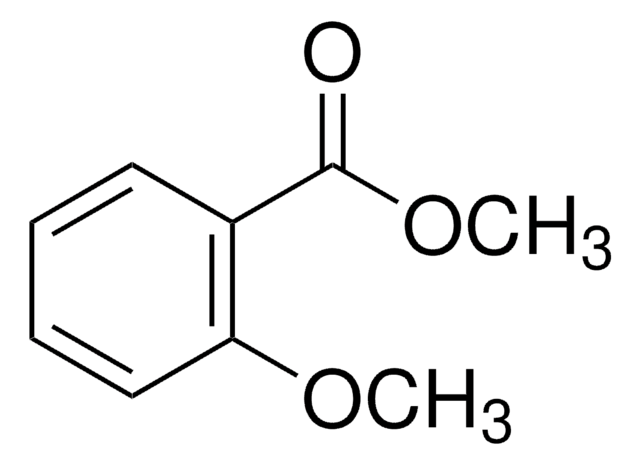
Methyl 3,5-dimethylbenzoate
98%
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula condensata:
(CH3)2C6H3CO2CH3
Numero CAS:
Peso molecolare:
164.20
Numero MDL:
Codice UNSPSC:
12352100
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Livello qualitativo
Saggio
98%
Forma fisica
solid
P. eboll.
239-240 °C (lit.)
Punto di fusione
31-33 °C (lit.)
Densità
1.027 g/mL at 25 °C (lit.)
Gruppo funzionale
ester
Stringa SMILE
COC(=O)c1cc(C)cc(C)c1
InChI
1S/C10H12O2/c1-7-4-8(2)6-9(5-7)10(11)12-3/h4-6H,1-3H3
PEVXENGLERTHJE-UHFFFAOYSA-N
Categorie correlate
Descrizione generale
Methyl 3,5-dimethylbenzoate is an aromatic carboxylic acid ester. It was selected as ligand during monomer screening for the synthesis and investigation of various europium compounds containing pinacolyl methylphosphonate with different ligands. Methyl 3,5-dimethylbenzoate is reported as precursor of methyl-3,5-divinylbenzoate.
Applicazioni
Methyl 3,5-dimethylbenzoate may be used in the preparation of four carbon isostere related to highly active 4-pyridinemethanols, which were subsequently evaluated for their antimalarial activity. It may be used in the total synthesis of (±)-indoxamycin B.
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
224.6 °F - closed cup
Punto d’infiammabilità (°C)
107.00 °C - closed cup
Dispositivi di protezione individuale
Eyeshields, Gloves, type N95 (US)
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
Oliver F Jeker et al.
Angewandte Chemie (International ed. in English), 51(14), 3474-3477 (2012-02-22)
Revised version: the first total synthesis of indoxamycin B leads to a stereochemical reassignment of the natural product. The synthetic route features an efficient carboannulation sequence to rapidly access the dihydroindenone system. Moreover, a series of Au(I)-catalyzed transformations served in
Molecularly imprinted polymers for the selective sequestering and sensing of ions.
The Johns Hopkins Medical Letter Health After 50, 18(4), 465-465 (1997)
A Markovac et al.
Journal of medicinal chemistry, 23(11), 1198-1201 (1980-11-01)
Four carbon isosteres related to the highly active 4-pyridylcarbinolamines were prepared and evaluated for suppressive antimalarial activity against Plasmodium berghei in mice. Three of the four examples possessed significant activity but were approximately one dose level less active than the
A L Jenkins et al.
Analytical chemistry, 71(2), 373-378 (1999-02-09)
The techniques of molecular imprinting and sensitized lanthanide luminescence have been combined to create the basis for a sensor that can selectively measure the hydrolysis product of the nerve agent Soman in water. The sensor functions by selectively and reversibly
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.