214027
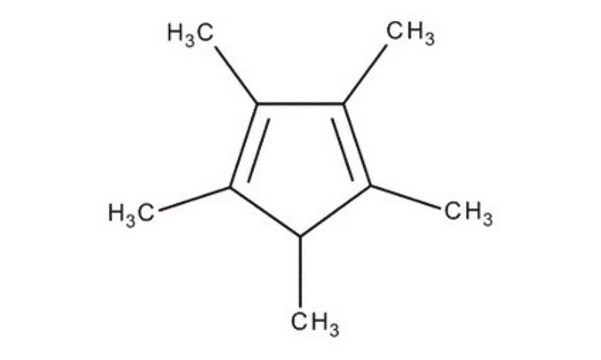
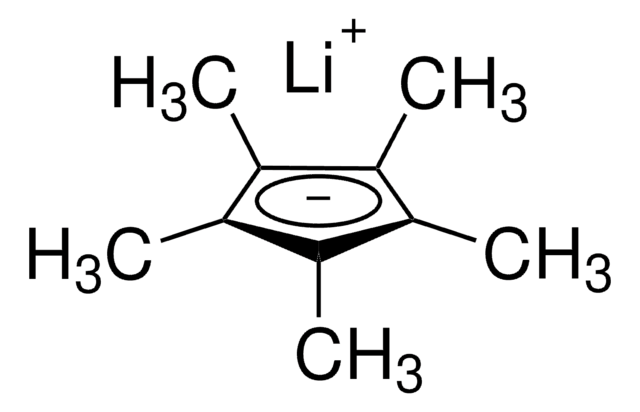
1,2,3,4,5-Pentamethylcyclopentadiene
95%
Sinonimo/i:
1,2,3,4,5-Pentamethyl-1,3-cyclopentadiene
About This Item
Prodotti consigliati
Livello qualitativo
Saggio
95%
Indice di rifrazione
n20/D 1.474 (lit.)
P. eboll.
58 °C/13 mmHg (lit.)
Densità
0.87 g/mL at 25 °C (lit.)
Stringa SMILE
CC1C(C)=C(C)C(C)=C1C
InChI
1S/C10H16/c1-6-7(2)9(4)10(5)8(6)3/h6H,1-5H3
WQIQNKQYEUMPBM-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Descrizione generale
Applicazioni
- Growth modifier chemical, during metal organic chemical vapour deposition of iron from iron pentacarbonyl.
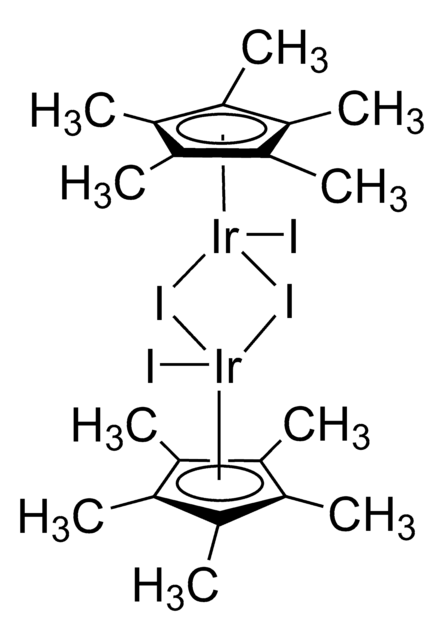
- Ligand in "one-pot" iridium-catalyzed transformation of alcohols to amides via the intermediacy of oximes.
- Raw material for the synthesis of [Cp*Rh(bpy)H2O]2+ (Cp* = pentamethylcyclopentadienyl, bpy = 2,2′-bipyridyl), an electron mediator in the regeneration process of NADH.
Avvertenze
Warning
Indicazioni di pericolo
Classi di pericolo
Flam. Liq. 3
Codice della classe di stoccaggio
3 - Flammable liquids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
111.2 °F - closed cup
Punto d’infiammabilità (°C)
44 °C - closed cup
Dispositivi di protezione individuale
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
Articoli
The Diels–Alder reaction is the reaction between a conjugated diene and an alkene (dienophile) to form unsaturated six-membered rings. Since the reaction involves the formation of a cyclic product via a cyclic transition state, it is also referred to as a "cycloaddition".
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.