213993
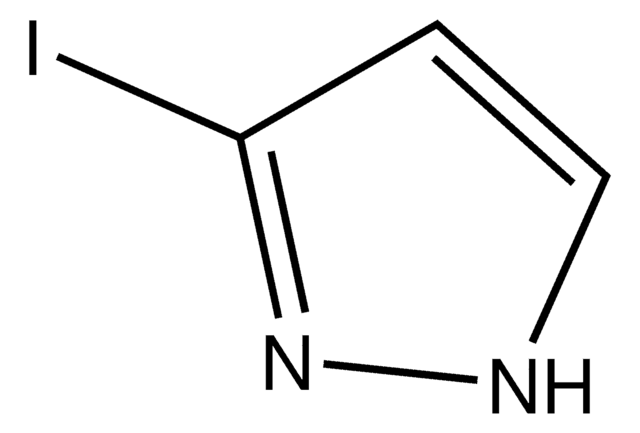
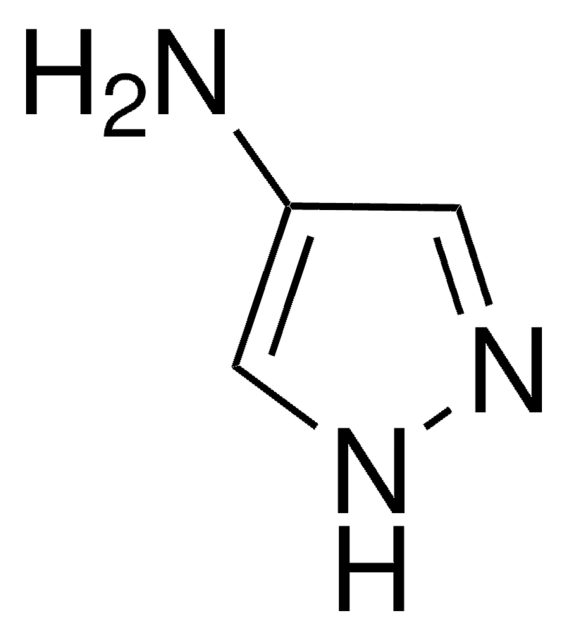
4-Iodopyrazole
99%
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula empirica (notazione di Hill):
C3H3IN2
Numero CAS:
Peso molecolare:
193.97
Numero CE:
Numero MDL:
Codice UNSPSC:
12352100
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Livello qualitativo
Saggio
99%
Forma fisica
solid
Punto di fusione
108-110 °C (lit.)
Gruppo funzionale
iodo
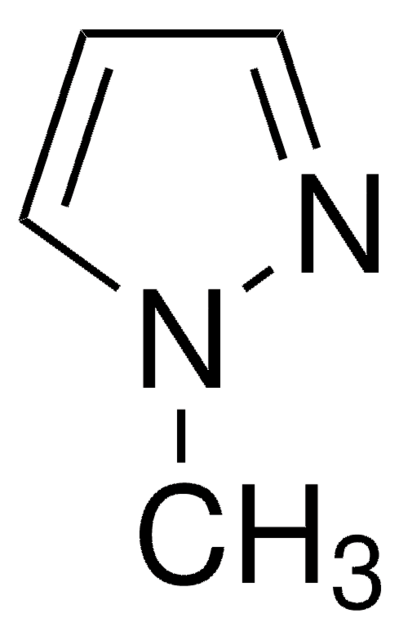
Stringa SMILE
Ic1cn[nH]c1
InChI
1S/C3H3IN2/c4-3-1-5-6-2-3/h1-2H,(H,5,6)
LLNQWPTUJJYTTE-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Descrizione generale
4-Iodopyrazole is a valuable intermediate for the synthesis of biologically active compounds. It undergoes iodination in the presence of iodine and ammonium hydroxide to yield 3,4-di-iodo- and 3,4,5-tri-iodo-pyrazole.
Applicazioni
4-Iodopyrazole was used in an indium-mediated synthesis of heterobiaryls.
Avvertenze
Warning
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Organi bersaglio
Respiratory system
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
dust mask type N95 (US), Eyeshields, Gloves
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Metin Zora et al.
The Journal of organic chemistry, 76(16), 6726-6742 (2011-07-12)
Electrophilic cyclizations of α,β-alkynic hydrazones by molecular iodine were investigated for the synthesis of 4-iodopyrazoles. α,β-Alkynic hydrazones were readily prepared by the reactions of hydrazines with propargyl aldehydes and ketones. When treated with molecular iodine in the presence of sodium
A Kojo et al.
Biochemical pharmacology, 42(9), 1751-1759 (1991-10-09)
Pyrazole and several of its derivatives increase the hepatic microsomal coumarin 7-hydroxylase to a variable extent. The strongest inducers are pyrazole itself and those derivatives which have a hydroxy group or a halogen at the 4-position of the molecule. The
Green iodination of pyrazoles with iodine/hydrogen peroxide in water.
Kim MM, et al.
Tetrahedron Letters, 49(25), 4026-4028 (2008)
Enrique Font-Sanchis et al.
The Journal of organic chemistry, 72(9), 3589-3591 (2007-04-05)
The palladium-mediated coupling reaction between triorganoindium reagents and organic electrophiles is extended to the synthesis of heteroaromatic compounds. Both electron-rich and electron-poor heterocycles can act as the organic electrophile or as the organoindium derivative.
Some iodinated pyrazole derivatives.
D Giles et al.
Journal of the Chemical Society. Perkin transactions 1, 13, 1179-1184 (1966-01-01)
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.