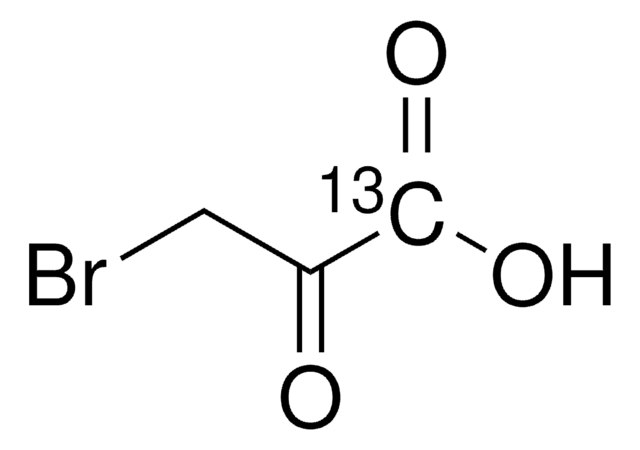
16490
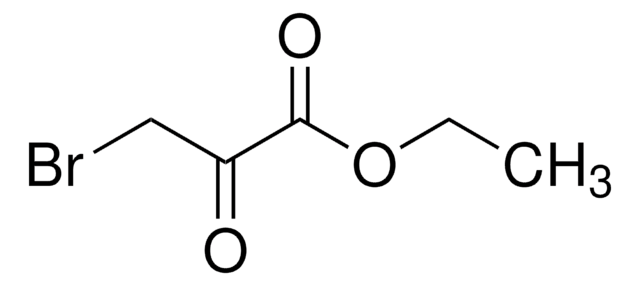
Bromopyruvic acid
≥98.0%
Sinonimo/i:
3-Bromo-2-oxopropionic acid
About This Item
Prodotti consigliati
Livello qualitativo
Saggio
≥98.0%
Forma fisica
(Powder or Crystals or Flakes)
Punto di fusione
77-82 °C
Solubilità
water: soluble 1 g/10 mL, clear to very slightly hazy, colorless
Gruppo funzionale
bromo
carboxylic acid
ketone
Temperatura di conservazione
2-8°C
Stringa SMILE
OC(=O)C(=O)CBr
InChI
1S/C3H3BrO3/c4-1-2(5)3(6)7/h1H2,(H,6,7)
PRRZDZJYSJLDBS-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Descrizione generale
Applicazioni
Altre note
Avvertenze
Danger
Indicazioni di pericolo
Classi di pericolo
Eye Dam. 1 - Skin Corr. 1B
Codice della classe di stoccaggio
8A - Combustible corrosive hazardous materials
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Articoli
We presents an article about the Warburg effect, and how it is the enhanced conversion of glucose to lactate observed in tumor cells, even in the presence of normal levels of oxygen. Otto Heinrich Warburg demonstrated in 1924 that cancer cells show an increased dependence on glycolysis to meet their energy needs, regardless of whether they were well-oxygenated or not.
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.