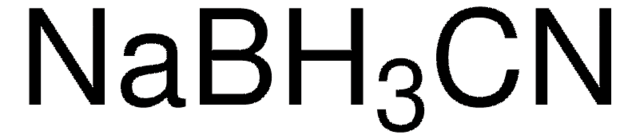06627
2-Aminoacridone
BioReagent, suitable for fluorescence, ≥98.0% (HPLC)
Synonyme(s) :
2-Amino-9(10H)-acridinone, 2-amino-10H-acridin-9-one, AMAC
About This Item
Produits recommandés
Gamme de produits
BioReagent
Niveau de qualité
Pureté
≥98.0% (HPLC)
Solubilité
DMF: soluble
DMSO: soluble
Fluorescence
λex 420 nm; λem 542 nm in 0.1 M Tris pH 8.0
Adéquation
suitable for fluorescence
Chaîne SMILES
Nc1ccc2Nc3ccccc3C(=O)c2c1
InChI
1S/C13H10N2O/c14-8-5-6-12-10(7-8)13(16)9-3-1-2-4-11(9)15-12/h1-7H,14H2,(H,15,16)
Clé InChI
PIGCSKVALLVWKU-UHFFFAOYSA-N
Vous recherchez des produits similaires ? Visite Guide de comparaison des produits
Catégories apparentées
Application
Conditionnement
Mention d'avertissement
Warning
Mentions de danger
Conseils de prudence
Classification des risques
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Organes cibles
Respiratory system
Code de la classe de stockage
11 - Combustible Solids
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Équipement de protection individuelle
dust mask type N95 (US), Eyeshields, Gloves
Certificats d'analyse (COA)
Recherchez un Certificats d'analyse (COA) en saisissant le numéro de lot du produit. Les numéros de lot figurent sur l'étiquette du produit après les mots "Lot" ou "Batch".
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
Articles
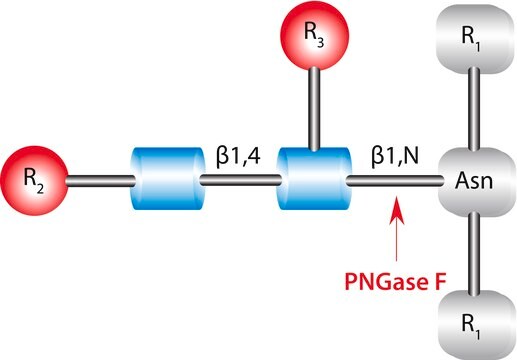
Structural modifications of proteins are essential to living cells. When aberrantly regulated they are often the basis of disease. Glycans are responsible for much of the structural variation in biologic systems, and their representation on cell surfaces is commonly called the “glycome.”
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique