302295
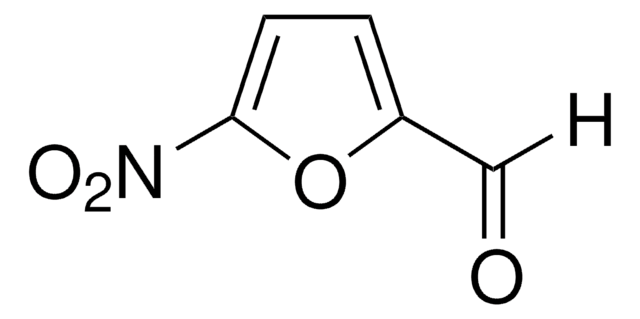
5-Nitro-2-thiophenecarboxaldehyde
98%
Se connecterpour consulter vos tarifs contractuels et ceux de votre entreprise/organisme
About This Item
Formule empirique (notation de Hill):
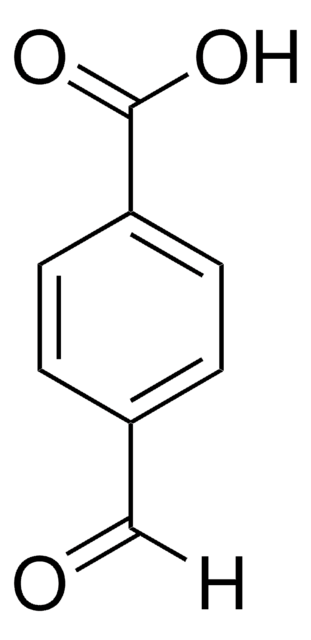
C5H3NO3S
Numéro CAS:
Poids moléculaire :
157.15
Numéro CE :
Numéro MDL:
Code UNSPSC :
12352100
ID de substance PubChem :
Nomenclature NACRES :
NA.22
Produits recommandés
Niveau de qualité
Pureté
98%
Forme
solid
Pf
75-77 °C (lit.)
Solubilité
acetone: soluble 1%, clear, yellow
Groupe fonctionnel
aldehyde
nitro
Chaîne SMILES
[H]C(=O)c1ccc(s1)[N+]([O-])=O
InChI
1S/C5H3NO3S/c7-3-4-1-2-5(10-4)6(8)9/h1-3H
Clé InChI
CHTSWZNXEKOLPM-UHFFFAOYSA-N
Catégories apparentées
Description générale
Diastereoselectivity in [4+2] cycloaddition of 1-methoxy-2-methyl-3-(trimethylsiloxy)-1,3-pentadiene with 5-nitro-2-thiophenecarboxaldehyde was investigated.
Application
5-Nitro-2-thiophenecarboxaldehyde was used in preparation of 2, 3-dihydro-2-(5-nitro-2-thienyl) quinazolin-4-(1H)-ones and various novel oxime ether derivatives, anti-protozoan agents.
Mention d'avertissement
Warning
Mentions de danger
Conseils de prudence
Classification des risques
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Organes cibles
Respiratory system
Code de la classe de stockage
11 - Combustible Solids
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Équipement de protection individuelle
dust mask type N95 (US), Eyeshields, Gloves
Faites votre choix parmi les versions les plus récentes :
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
Michael P Doyle et al.
Proceedings of the National Academy of Sciences of the United States of America, 101(15), 5391-5395 (2004-04-03)
Chiral dirhodium(II) carboxamidates are highly efficient catalysts for reactions between a variety of aldehydes and activated dienes. Catalyst loadings as low at 0.01 mol % have been realized with enantioselectivities up to 97%. Kinetic investigations reveal a pronounced electronic influence
Antibacterial 2,3-dihydro-2-(5-nitro-2-thienyl)-quinazolin-4(1H)-ones.
R J Alaimo et al.
Journal of medicinal chemistry, 15(3), 335-336 (1972-03-01)
Synthesis and< i> in vitro</i> anti-protozoan activity of new 5-nitrothiophene oxime ether derivatives.
Delmas F, et al.
European Journal of Medicinal Chemistry, 28(1), 23-27 (1993)
Ali Almasirad et al.
Iranian journal of pharmaceutical research : IJPR, 10(4), 727-731 (2011-10-01)
A series of new 2-(phenylthio) benzoylarylhydrazones were synthesized by acid-catalyzed condensation of hydrazide 3 with corresponding aldehydes. The chemical structures of the compounds were elucidated by FT-IR, (1)H-NMR and Mass spectra. All newly synthesized compounds were evaluated for their antimycobacterial
Jian Xu et al.
Food chemistry, 221, 1530-1538 (2016-12-17)
We synthesized a series of 4- or 5-functionalized TCT derivatives (1-12) and investigated their inhibitory activities and mechanisms on tyrosinase by using Spectrofluorimetry, 1H and 13C NMR titration and IR spectra. The results of the fluorescence spectra and NMR titrations
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique