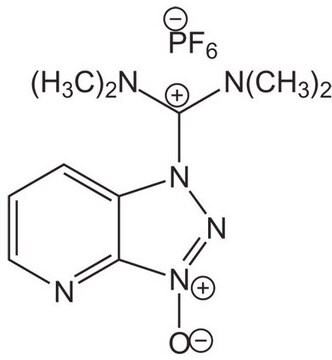
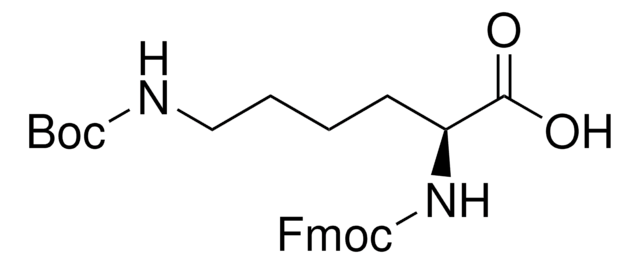
04936
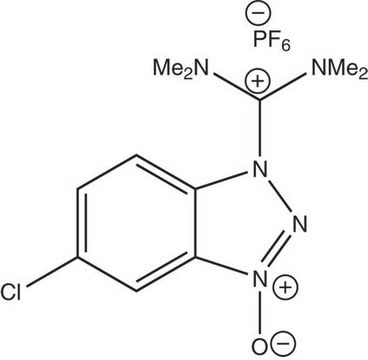
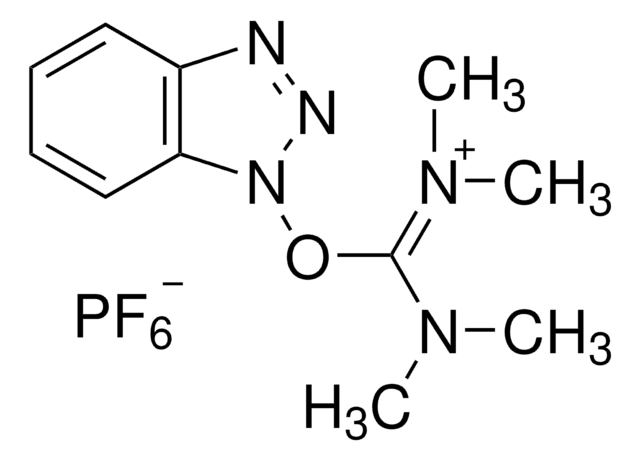
O-(6-Chlorbenzotriazol-1-yl)-N,N,N′,N′-tetramethyluronium-hexafluorphosphat
≥98.0% (HPLC)
Synonym(e):
1-[Bis-(dimethylamino)-methylen]-5-chlor-benzotriazolium-3-oxid-hexafluorophosphat, N,N,N′,N′-Tetramethyl-O-(6-chlor-1H-benzotriazol-1-yl)-uronium-hexafluorphosphat, HCTU
About This Item
Empfohlene Produkte
Qualitätsniveau
Assay
≥98.0% (HPLC)
Form
powder
Eignung der Reaktion
reaction type: Coupling Reactions
Verunreinigungen
≤0.5% water
mp (Schmelzpunkt)
185-190 °C
Anwendung(en)
peptide synthesis
SMILES String
ClC1=CC=C(N([C+](N(C)C)N(C)C)N=[N+]2[O-])C2=C1.F[P-](F)(F)(F)(F)F
InChI
1S/C11H15ClN5O.F6P/c1-15(2)11(16(3)4)18-17-10-7-8(12)5-6-9(10)13-14-17;1-7(2,3,4,5)6/h5-7H,1-4H3;/q+1;-1
InChIKey
ZHHGTMQHUWDEJF-UHFFFAOYSA-N
Anwendung
Synthesis of near-infrared pH activatable fluorescent probes
Synthesis of human β-amyloid by Fmoc chemistry
Stereoselective Horner-Wadsworth-Emmons olefination
Covalent ligation of fluorescent peptides to quantum dots
Alkylation of human telomere sequence
Sonstige Hinweise
Signalwort
Warning
H-Sätze
Gefahreneinstufungen
Skin Sens. 1A
Lagerklassenschlüssel
11 - Combustible Solids
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Kunden haben sich ebenfalls angesehen
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.