U211140
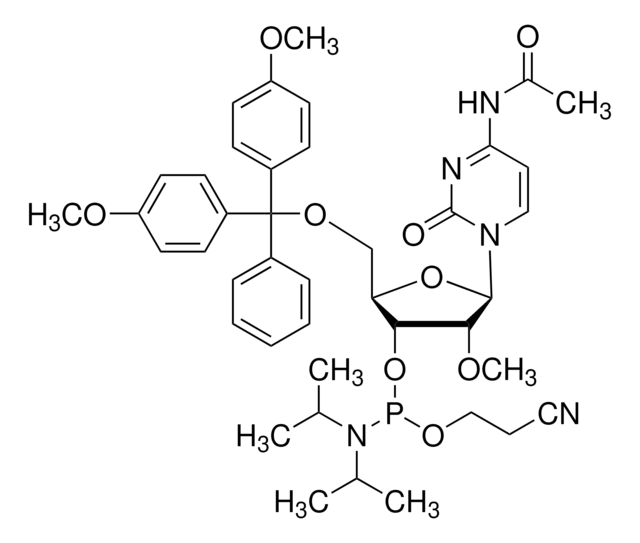
DMT-2′O-Methyl-rU Phosphoramidite
configured for MerMade
Sinônimo(s):
5′-O-[bis(4-methoxyphenyl)phenylmethyl]-2′-O-methyl-uridine, 3′-[2-cyanoethyl N,N-bis(1-methylethyl)phosphoramidite], DMT-2′O-Methyl-rU amidite
About This Item
Produtos recomendados
fonte biológica
non-animal source (no BSE/TSE risk)
Nível de qualidade
linha de produto
Proligo Reagents
Ensaio
≥99% (31P-NMR)
≥99.0% (reversed phase HPLC)
Formulário
powder
peso molecular
760.81 g/mol
Impurezas
≤0.3% mU2 (reversed phase HPLC, Hydrolysate)
≤0.3% mU3 (reversed phase HPLC, DMT-rme)
≤0.3% water content (Karl Fischer)
≤0.5% P(III) Impurities 100-169ppm (31P-NMR)
≤0.5% single Impurity (redox titration)
≤1.0% mU1 (reversed phase HPLC, DMT-rUme-DMT)
≤3% residual Solvent content
cor
white to off-white
adequação
conforms to structure for H-NMR
conforms to structure for LC-MS
compatibilidade
configured for MerMade
temperatura de armazenamento
2-8°C
cadeia de caracteres SMILES
CO[C@@H]1[C@H](OP(OCCC#N)N(C(C)C)C(C)C)[C@@H](COC(c2ccccc2)(c3ccc(OC)cc3)c4ccc(OC)cc4)O[C@H]1N5C=CC(=O)NC5=O
InChI
1S/C40H49N4O9P/c1-27(2)44(28(3)4)54(51-25-11-23-41)53-36-34(52-38(37(36)49-7)43-24-22-35(45)42-39(43)46)26-50-40(29-12-9-8-10-13-29,30-14-18-32(47-5)19-15-30)31-16-20-33(48-6)21-17-31/h8-10,12-22,24,27-28,34,36-38H,11,25-26H2,1-7H3,(H,42,45,46)/t34-,36-,37-,38-,54?/m1/s1
chave InChI
UVUOJOLPNDCIHL-XKZJCBTISA-N
Procurando produtos similares? Visita Guia de comparação de produtos
Descrição geral
- High yield of crude oligonucleotides Compatible with DNA synthesis
- Can be employed together with DNA or RNA phosphoramidites in the same synthesis to produce mixmer oligonucleotides
- Recommended deprotection conditions are 8 hours at 55 deg C using concentrated ammonia solution, or with AMA (concentrated ammonia/40% aqueous methylamine I/I, v/v) for 10 minutes at 65 deg C
- Purification and other downstream processing of fully modified 2′OMethyl RNA oligonucleotides are simpler than in the case of RNA, as no special precautions are required to provide protection against nucleolytic degradation
- Synthesis of 2′O-Methyl RNA oligonucleotides is similar to standard DNA synthesis but requires an elongated coupling time (recommended is 6minutes compared to 90 seconds for DNA monomers)
- 2′O-Methyl RNA phosphoramidites are also available with fast deprotection chemistry
Aplicação
- Diagnostic probes
- Aptamer and ribozyme development
- Mixed 2′O-Methyl-RNA/DNA antisense molecules
Código de classe de armazenamento
11 - Combustible Solids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica