529311
Iron(III) oxide
≥99.995% trace metals basis
Sinônimo(s):
Ferric oxide
About This Item
Produtos recomendados
Nível de qualidade
Ensaio
≥99.995% trace metals basis
forma
powder
aplicação(ões)
battery manufacturing
cadeia de caracteres SMILES
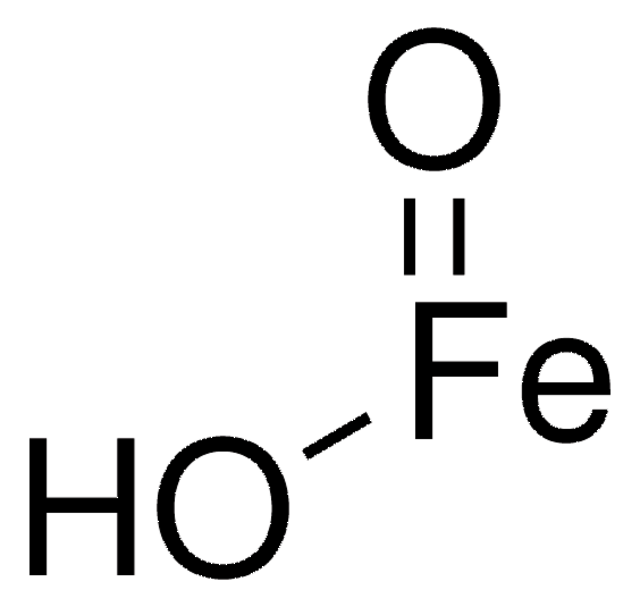
O=[Fe]O[Fe]=O
InChI
1S/2Fe.3O
chave InChI
JEIPFZHSYJVQDO-UHFFFAOYSA-N
Procurando produtos similares? Visita Guia de comparação de produtos
Descrição geral
Aplicação
Código de classe de armazenamento
13 - Non Combustible Solids
Classe de risco de água (WGK)
nwg
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Equipamento de proteção individual
dust mask type N95 (US), Eyeshields, Gloves
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
Artigos
Magnetism and magnetic materials have been of scientific interest for over 1,000 years. More recently, fundamental investigations have focused on exploring the various types of magnetic materials and understanding the magnetic effects created by electric currents.
Magnetic materials permeate numerous daily activities in our lives. They are essential components of a diversity of products including hard drives that reliably store information on our computers, decorative magnets that keep the shopping list attached to the refrigerator door, electric bicycles that speed our commute to work, as well as wind turbines for conversion of wind energy to electrical power.
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica