About This Item
Fórmula empírica (Notação de Hill):
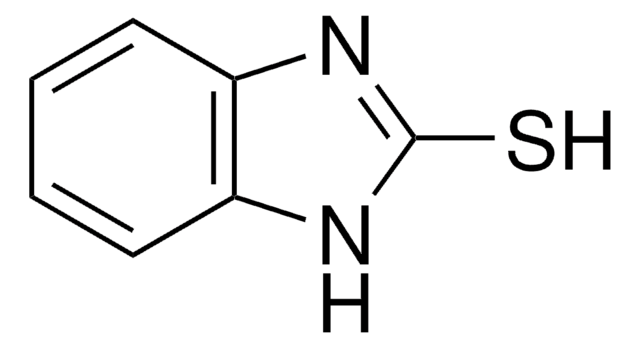
C7H4OS2
Número CAS:
Peso molecular:
168.24
Beilstein:
119513
Número CE:
Número MDL:
Código UNSPSC:
12352100
ID de substância PubChem:
NACRES:
NA.22
Produtos recomendados
Nível de qualidade
Ensaio
97%
forma
solid
pf
74-77 °C (lit.)
solubilidade
toluene: soluble 2.5%, clear, yellow
grupo funcional
disulfide
cadeia de caracteres SMILES
O=C1SSc2ccccc12
InChI
1S/C7H4OS2/c8-7-5-3-1-2-4-6(5)9-10-7/h1-4H
chave InChI
GZTYTTPPCAXUHB-UHFFFAOYSA-N
Procurando produtos similares? Visita Guia de comparação de produtos
Categorias relacionadas
Descrição geral
3H-1,2-Benzodithiol-3-one is a heterocyclic building block.
3H-1,2-Benzodithiol-3-one is reported to react with triphenylphosphine to afford dithiosalicylide.
Aplicação
3H-1,2-Benzodithiol-3-one (1,2-benzodithiol-3-one) may be used as sulfur-transferring agent for the transformation of H-phosphonothioate and H-phosphonate diesters into the corresponding phosphorodi- and phosphoromonothioates.
3H-1,2-Benzodithiol-3-one may be employed as sulfurizing reagent in the preparation of phosphorothioate-containing oligodeoxyribonucleotides.
Código de classe de armazenamento
11 - Combustible Solids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Equipamento de proteção individual
Eyeshields, Gloves, type N95 (US)
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Q Xu et al.
Nucleic acids research, 24(9), 1602-1607 (1996-05-01)
Previous methods for the preparation of phosphorothioate-containing oligodeoxyribonucleotides rely on the reaction of phosphite triesters with sulfurizing reagents such as tetraethylthiuram disulfide (TETD) and 3H-1,2-benzodithiol-3-one 1,1-dioxide (Beaucage reagent). However, these and other sulfurizing reagents suffer from several disadvantages, and there
Nucleoside H-phosphonates. 13. Studies on 3H-1, 2-benzodithiol-3-one derivatives as sulfurizing reagents for H-phosphonate and H-phosphonothioate diesters.
Stawinski J and Thelin M.
The Journal of Organic Chemistry, 56(17), 5169-5175 (1991)
Novel syntheses of dithiosalicylide.
Mitra K and Gates KS.
Tetrahedron Letters, 36(9), 1391-1394 (1995)
Santhosh Sivaramakrishnan et al.
Bioorganic & medicinal chemistry letters, 18(10), 3076-3080 (2007-12-11)
Though less potent than the parent natural product leinamycin, S-deoxyleinamycin displays activity against human cancer cell lines that is comparable to many clinically used agents. The results reported here suggest that the 1,2-dithiolan-3-one heterocycle found in S-deoxyleinamycin reacts with thiols
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica