P8465
Protease Inhibitor Cocktail
lyophilized powder, for the inhibition of serine, cysteine, aspartic, metalloproteases and aminopeptidases, for use with bacterial cell extracts, lyophilized powder
Synonyme(s) :
Protease inhibitor
About This Item
Produits recommandés
product name
Protease Inhibitor Cocktail powder, for use with bacterial cell extracts, lyophilized powder
Niveau de qualité
Forme
lyophilized powder
Température de stockage
−20°C
Vous recherchez des produits similaires ? Visite Guide de comparaison des produits
Description générale
The product contains individual components that target serine, cysteine, aspartic, and metalloproteases, as well as aminopeptidases.
Application
Actions biochimiques/physiologiques
Caractéristiques et avantages
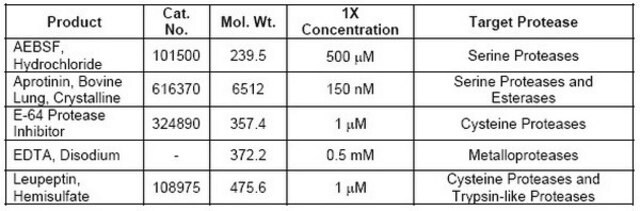
Contains individual components, including AEBSF, EDTA, Bestatin, Pepstatin A, and E-64, each targeting specific types of proteases.
Lyophilized powder is stable for at least 2 years when stored unopened at -20°C.
Supplied with a vial of DMSO for preparation of a cocktail solution.
One mL of the cocktail solution is recommended for the inhibition of protease activity found in 20 mL of cell lysate from 4g of E. coli cells.
Attention
Notes préparatoires
Produit(s) apparenté(s)
Mention d'avertissement
Danger
Mentions de danger
Classification des risques
Acute Tox. 4 Inhalation - Eye Dam. 1 - Met. Corr. 1 - Skin Corr. 1A - STOT RE 2 Inhalation
Organes cibles
Respiratory Tract
Code de la classe de stockage
8A - Combustible corrosive hazardous materials
Classe de danger pour l'eau (WGK)
WGK 3
Équipement de protection individuelle
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
Certificats d'analyse (COA)
Recherchez un Certificats d'analyse (COA) en saisissant le numéro de lot du produit. Les numéros de lot figurent sur l'étiquette du produit après les mots "Lot" ou "Batch".
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique