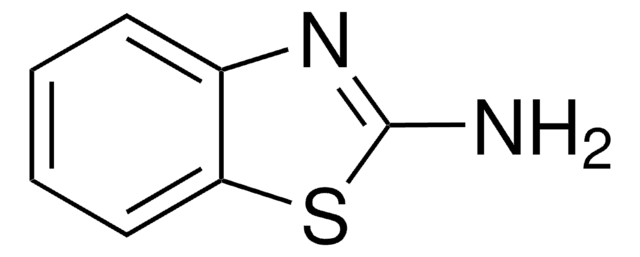
180459
5,6,7,8-Tetrahydro-2-naphthol
98%
Se connecterpour consulter vos tarifs contractuels et ceux de votre entreprise/organisme
About This Item
Formule empirique (notation de Hill):
C10H12O
Numéro CAS:
Poids moléculaire :
148.20
Numéro CE :
Numéro MDL:
Code UNSPSC :
12352100
ID de substance PubChem :
Nomenclature NACRES :
NA.22
Produits recommandés
Pureté
98%
Point d'ébullition
275-276 °C (lit.)
Pf
59-61 °C (lit.)
Chaîne SMILES
Oc1ccc2CCCCc2c1
InChI
1S/C10H12O/c11-10-6-5-8-3-1-2-4-9(8)7-10/h5-7,11H,1-4H2
Clé InChI
UMKXSOXZAXIOPJ-UHFFFAOYSA-N
Catégories apparentées
Application
5,6,7,8-Tetrahydro-2-naphthol was used as a model compound in the study of photochemical transformation of 17β-estradiol (natural estrogenic steroid) and 17α-ethinylestradiol (synthetic oral contraceptive).
Mention d'avertissement
Warning
Mentions de danger
Conseils de prudence
Classification des risques
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Organes cibles
Respiratory system
Code de la classe de stockage
11 - Combustible Solids
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
235.4 °F - closed cup
Point d'éclair (°C)
113 °C - closed cup
Équipement de protection individuelle
dust mask type N95 (US), Eyeshields, Gloves
Faites votre choix parmi les versions les plus récentes :
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
T Bhattacharya et al.
Spectrochimica acta. Part A, Molecular and biomolecular spectroscopy, 59(3), 525-535 (2003-01-14)
Both steady state and time resolved spectroscopic measurements reveal that the prime process involved in quenching mechanism of the lowest excited singlet (S1) and triplet (T1) states of the well known electron acceptor 9-Cyanoanthracene (9CNA) in presence of 5,6,7,8-tetrahydro-1-naphthol (TH1N)
B Kalyanaraman et al.
The Journal of biological chemistry, 259(22), 14018-14022 (1984-11-25)
Electron spin resonance spectroscopy has been used to demonstrate production of semiquinone-free radicals from the oxidation of the catechol estrogens 2- and 4-hydroxyestradiol and 2,6- and 4,6-dihydroxyestradiol. Radicals were generated either enzymatically (using horseradish peroxidase-H2O2 or tyrosinase-O2) or by autoxidation
Patrick Mazellier et al.
Chemosphere, 73(8), 1216-1223 (2008-09-03)
The photochemical transformation of natural estrogenic steroid 17beta-estradiol (E2) and the synthetic oral contraceptive 17alpha-ethinylestradiol (EE2) has been studied in dilute non buffered aqueous solution (pH 5.5-6.0) upon monochromatic (254 nm) and polychromatic (lambda>290 nm) irradiation. Upon irradiation at 254
N M Howarth et al.
Steroids, 62(4), 346-350 (1997-04-01)
In our continuing quest to design efficient inhibitors of estrone sulfatase activity and to assess the recognition of estrone sulfate surrogates by estrone sulfatase, we synthesized and evaluated several sulfonate derivatives of 5,6,7,8-tetrahydronaphth-2-ol and estrone. 5,6,7,8-Tetrahydronaphth-2-methanesulfonate (11), and 5,6,7,8-tetrahydronaphth-2-(p-toluene)sulfonate (12)
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique
![[1,1′-Bis(diphénylphosphino)ferrocène]dichloropalladium(II), complexe avec le dichlorométhane](/deepweb/assets/sigmaaldrich/product/structures/825/986/4317978b-1256-4c82-ab74-6a6a3ef948b1/640/4317978b-1256-4c82-ab74-6a6a3ef948b1.png)

![[1,1′-bis(diphénylphosphino)ferrocène]dichloropalladium(II)](/deepweb/assets/sigmaaldrich/product/structures/130/734/8846aa26-1858-458a-998d-8c306c13bf0f/640/8846aa26-1858-458a-998d-8c306c13bf0f.png)